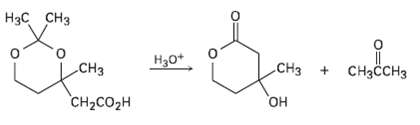
The following reaction involves a hydrolysis followed by an intra molecular nucleophilic acyl substitution reaction. Write both
Question:
The following reaction involves a hydrolysis followed by an intra molecular nucleophilic acyl substitution reaction. Write both steps, and show theirmechanisms.
Transcribed Image Text:
Нзс снз смдон CHз CHз H30* CHзсCHз CH2CO2H Он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
This mechanism divides into two sequences of steps In the first part of the ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reaction involves an intra molecular Michael reaction followed by an intra molecular aldol reaction. Write both steps, and show theirmechanisms. NaOH Ethanol
-
The following reaction involves an intra molecular aldol reaction followed by a ret ro aldol-like reaction. Write both steps, and show theirmechanisms. O2Et CO2Et Na* "OEt Ethanol
-
The following reaction involves two successive intra molecular Michael reactions. Write both steps, and show theirmechanisms. Nat "OEt Ethanol
-
Samson Company manufactures embroidered jackets. The company uses a standard cost system to control manufacturing costs. The following data represent the standard unit cost of a jacket: Fixed...
-
In the United States, most hospitals are non-profit but nearly all pharmaceutical firms are for profit. Can you offer an explanation based on the consumer shopping problem?
-
A study was conducted at Virginia Tech to determine if certain static arm-strength measures have an influence on the dynamic lift characteristics of an individual. Twenty-five individuals were...
-
Explain medical staff organization and committee structure.
-
Kiel Centers sales are all made on account. The firms collection experience has been that 35% of a months sales are collected in the month the sale is made, 55% are collected in the month following...
-
Is the public debt (unbalanced budget) bad? Improve, adjust, or completely change your original answer by discussing the pros and cons of debt based on what we learned. For example, - What negative...
-
You have been given the following information about the production of Horatio Co., and are asked to provide the plant manager with information for a meeting with the vice president of operations. The...
-
The Darzens reaction involves a two-step, base-catalyzed condensation of ethyl chloroacetate with a ketone to yield an epoxy ester. The first step is a carbonyl condensation reaction, and the second...
-
The following reaction involves a conjugate addition reaction followed by an intra molecular Claisen condensation. Write both steps, and show theirmechanisms. CO2CH3 (CH3i>CuLi -CEC-CO2CH3 H
-
Describe why book value per share of common stock may not be useful for investment analysis.
-
Margaret works at a CPA firm because she needs tables; she hates numbers and accountants. She then wants to rent these tables at horror conventions. She loves her tables. Choose the true statements)....
-
For taxpayers under the Large Corporate Compliance Program, the IRS will typically gather evidence by issuing: a. Information document requests b. Pre-summons letters C. Summons d. Designated Summons...
-
Un empleado se encuentra insatisfecho con la resolucin de un conflicto tico en su lugar de empleo. De acuerdo con el Instituto de Contadores Administrativos (Institute of Management Accountants), el...
-
Andrew owes Laila100,000 for the purchase of construction materials. Laila also owes Andrew a refund worth 20,000 for an earlier purchase made. Was there extinguishment of the obligations? a. Yes,...
-
On 12/12/2020 smith inc purchased merchandise on account from suppliers. On 12/20/2020 smith inc paid its suppliers for half of the merchandise purchased. Smith inc did not record either of these...
-
George has $91,700 in salary from his full-time position and $43,000 in net income in 2017 from his sole proprietorship. What is his self-employment tax? What portion of this can he deduct?
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
What is the electron sea model for bonding in metals?
-
Predict the products, including stereochemistry where appropriate, for the m-chloroperoxybenzoic acid epoxidations of the following alkenes. (a) cis-hex-2-ene (b) trans-hex-2-ene (c) cis-cyclodecene...
-
Predict the major products of the following reactions, and propose mechanisms to support your predictions. (a) (b) 1-methylcyclopentene HBr CH C-O-O-C-CH l-phenylpropene + HBr + di-tert-butyl...
-
(a) Propose a mechanism for the conversion of cis-hex-3-ene to the epoxide (3,4-epoxyhexane) and the ring-opening reaction to give the glycol, hexane-3,4-diol. In your mechanism, pay particular...
-
A couple obtained a $20,000 mortgage loan at an interest rate of 10.5% compounded monthly. (Original principal equals to PV of all payments discounted at the interest rate on the loan contract) (1)...
-
What strategies and tactics are employed to manage strategic risks and uncertainties, including geopolitical instability, supply chain disruptions, and emerging competitive threats, while preserving...
-
How do strategic planners integrate ethical considerations and sustainability imperatives into strategic planning processes, balancing short-term financial objectives with long-term societal and...

Study smarter with the SolutionInn App


