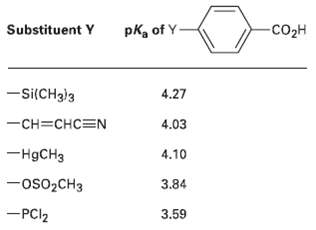
The pK a ?s of five p-substituted benzoic acids (YC 6 H 4 CO 2 H) follow.
Question:
The pKa?s of five p-substituted benzoic acids (YC6H4CO2H) follow. Rank the corresponding substituted benzenes (YC6H5) in order of their increasing reactivity toward electrophilic aromatic substitution. If benzoic acid has PKa = 4.19, which of the sub-stituents are activators and which arc deactivators?
Transcribed Image Text:
-CO2H Substituent Y pka of Y- -si(CH3)3 4.27 -CH=CHC=N 4.03 -H9CH3 4.10 -OS02CH3 3.84 -PCI2 3.59
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (19 reviews)
Substituent PC1 OSOCH3 CHCHCN HgCH3 H pka 359 384 403 410 419 427 Acidity Most aci...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
List the following substances in order of their increasing oxidizing power: (a) Acetoacetate, (b) Cytochrome b (Fe3+), (c) NAD+ (d) SO24-, and (e) Pyruvate.
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
Rank the following compounds in order of increasing reactivity toward nitration with HNO3 and explain your choices: thiophene, benzene, 3-methylthiophene, and Z-methvlfuran.
-
You kick a soccer ball, compressing it suddenly to 2/3 of its original volume. In the process, you do 410 J of work on the air (assumed to be an ideal gas) inside the ball. (a) What is the change in...
-
1. Does Louie have a problem, or are the people who made the negative comments about Louie just being too sensitive? 2. What improvements might Louie need to make to become a truly multicultural...
-
A hot steam pipe having an inside surface temperature of 250C has an inside diameter of 8 cm and a wall thickness of 5.5 mm. It is covered with a 9-cm layer of insulation having k = 0.5 W/m C,...
-
The following plot of the voltage output from a VFD illustrates why the total 600 V is always present in the common-mode voltage from the source. What is the common-mode voltage of utility power? 0.5...
-
The partnership of Hendrick, Mitchum, and Redding has the following account balances: Cash . . . . . . . . . . . . . . . . . . . . . . $ 50,000 Noncash assets . . . . . . . . . . . . . . .135,000...
-
During 20--, Rachael Lopez, president of Mathieson Company, was paid a semimonthly salary of $9,825. Compute the amount of FICA taxes that should be withheld from her: a. 9th paycheck b. 17th...
-
The disclosure rules for business combinations complicate financial analysis. Trend analysis becomes difficult because comparative financial statements are not retroactively adjusted to include data...
-
Propose a synthesis of the anti-inflammatory drug Fenclorac fromphenyl-cyclohexane. CI Fenclorac CHCO2H
-
How would you carry out the following transformations? More than one step is required in each case. (a) H (b) Ph "Co "
-
The notation________is used to describe the fact that as x gets closer to c, but remains greater than c, the value of gets closer to R.
-
How can you satisfy whether a proprietary company is small or large? Justify your answer with points.?
-
What consideration may discourage executives from disclosing information about many segments of the enterprise?
-
I - Paul is the sole incorporator of Hemp Co., a company that processes hemp into clothing. Prior to incorporation, Paul enters into a contract on Hemp Co.'s behalf with a local farmer for the...
-
How might cash flow risk increase during difficult market environments?
-
What type of conflict is happening if two teams in the same accounting department have a dispute with each other?
-
Repeat Problem 23 assuming that the stock price is growing at 3 percent per year. Data from problem 23 Lahoma Enterprises, Inc., needs $15 million to finance a major product development. The project...
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
Which solid in each pair has the higher melting point and why? a. C. TiO (s) or HOOH(s) Kr(s) or Xe(s) b. CCl4(s) or SiCl4(s) d. NaCl(s) or CaO(s)
-
Estimate each of the bond angles and order the bond lengths (smallest first) for each of the following molecule. (a) a) (b) d) Br:
-
Estimate each of the bond angles and order the bond lengths (smallest first) for each of the following molecule. (a) a) (b) d) Br:
-
(a) Give the H-C=O bond angle in methyl formate. (b) One dihedral angle in methyl formate relates the plane containing the O=C-O bonds to the plane containing the C-O-C bonds. Sketch two structures...
-
Reflect on a group interview you've had: * Was there anything you struggled with in preparing for the peer interviews? * What would you have changed about your interview? What would you have kept the...
-
Please conduct comprehensive research on the status of Metaverse, Choose a company, an industry, a product, or a service, and Share the strategies being used in Metaverse to develop a competitive...
-
Renovation and Restoration of community Park project For Risk management plan External risk : Budget Fluctuations in local economy Environment disasters Internal Risk: Safety Not enough money, staff...

Study smarter with the SolutionInn App


