Question: The spectral, hemispherical absorptivity of an opaque surface is as shown. What is the solar absorptivity, ?S? If it is assumed that ?? = ??
The spectral, hemispherical absorptivity of an opaque surface is as shown. What is the solar absorptivity, ?S? If it is assumed that ?? = ?? and that the surface is at a temperature of 340 K. what is its total, hemispherical emissivity?
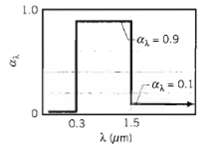
1.0 az = 0.9 - az=0.1 0.3 1.5 a um)
Step by Step Solution
★★★★★
3.30 Rating (162 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

KNOWN Spectral hemispherical absorptivity of an opaque surface FIND a Solar absorptivity b Total hem... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
8-E-M-E-H-M-T (1131).docx
120 KBs Word File


