The syntheses shown here are unlikely to occur as written. What is wrong witheach? 1. Mg 2.
Question:
The syntheses shown here are unlikely to occur as written. What is wrong witheach?
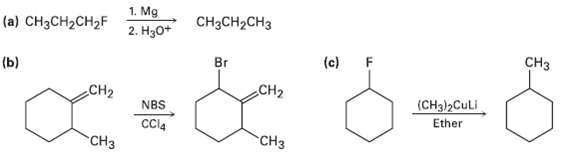
Transcribed Image Text:
1. Mg 2. Нзо* CH3CH2CH3 (a) CH3CH2CH2F Cнз (c) F CH2 Br (ь) CH2 (CH3)2CULI Ether NBS CCl4 "СНз CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
a Fluoroalkanes dont usually form Grignard reagen...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Here are five questions written by one of your systems analysis team members. Her interviewee is the local manager of LOWCO, an outlet of a national discount chain, who has asked you to work on a...
-
What is wrong with these synthesesexplain. 1) NaNH2, NH, (1) C=CCH3 a) CH3C=CH 2) -Br CH;CH,-NH, I b) CH;CH,I + NH3 CI + Br OCH, CH, + CH,O d) . .. Br + HBr e) CH3 CH3 H,SO, f)
-
8 for 0 < < 6 for 6
-
Refer to the Snoey Software Company case. Design a spreadsheet that will determine the annual profit when the prices for the Educational, Large-Scale, and Professional versions are $100, $300, and...
-
Use superposition to find Vo in the circuit of Fig. 4.86. 1 0.5 2 10 V 2 A
-
Magic Aqua Ltd, which runs competitive swimming schools for children aged 816, has budgeted revenue for the first 6 months of 2025 as follows. All revenue is provided on account and Magic Aqua Ltd...
-
Provide evidence to support the following statement: The growth of e-business is due to both business and technological innovations.
-
Explain how RTOS manage interrupt handling to minimize latency. What design considerations are typically employed to ensure low-latency interrupt processing ?
-
A retail Pet Store has asked you to design a database to capture the important aspects of their business data. In this assignment, you will build on the basic design to add tools to assist the user...
-
How would you carry out the followingsyntheses? Cyclohexene Cyclohexanol Cyclohexane ~/~/al
-
Why do you suppose it?s not possible to prepare a Grignard reagent from a bromo alcohol such as 4-bromo-1-pentanol? Give another example of a molecule that is unlikely to form a Grignard reagent....
-
You have just graduated from the MBA program of a large university, and one of your favorite courses was "Today's Entrepreneurs." In fact, you enjoyed it so much you have decided you want to "be your...
-
What amount of money invested today at 2.49% compounded semiannually will have an accumulated value of $809,500 in 7 years from now. P/Y = PV = $ C/Y = PMT= $ N= FV = $ I/Y = %
-
For calendar year 2022, Stuart and Pamela Gibson file a joint return reflecting AGI of $372,000. Their itemized deductions are as follows: Note: All expenses are before any applicable limitations,...
-
1. What year are you preparing your students for? 1973? 1995? Provide evidence that your school's curriculum and the program you use are preparing your students for 2030? Are you even preparing them...
-
Portsmouth Company makes upholstered furniture. Its only variable cost is direct materials. The demand for the company's products far exceeds its manufacturing capacity. The bottleneck (or...
-
1. Compute the lower of cost or market for the Inventory applied separately to each item. 2. If the market amount is less than the recorded cost of the inventory, then record the LCM adjustment to...
-
Do you think that a new CEO could have turned around the culture at Vice Media?
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
List the following gas-phase ion pairs in order of the quantity of energy released when they form from separated gas-phase ions. List the pair that releases the least energy first. Na + F , Mg 2+ F ...
-
Draw a reaction-energy diagram for the following reaction: CH3 + Cl2 CH3Cl + Cl The activation energy is 4 kJ mol (1 kcal mol), and the overall Ho for the reaction is -109kJ/mole (-26 kcal/mol) (b)...
-
The bromination of methane proceeds through the following steps: (a) Draw a complete reaction-energy diagram for this reaction. (b) Label the rate-limiting step. (c) Draw the structure of each...
-
(a) Using the BDEs in Table 4-2 (p. 143), compute the value of Ho for each step in the iodination of methane. (b) Compute the overall value of Ho for iodination. (c) Suggest two reasons why iodine...
-
2-Using the CPT data in figure below, a unit weight of 115 pcf, and an overconsolidation ratio of 3, determine (a) soil behavior type (b) o' using Kulhawy and Mayne correlation and (c) N60 for the...
-
How does the application of computational fluid dynamics (CFD) coupled with multiphase flow modeling enhance the understanding and prediction of complex hydrodynamic behaviors in industrial-scale...
-
1. From the joint distribution defined by the graphical model, determine if the conditional independence holds: ALL FIE, B, and provide brief explanation (5 points). 2. Given an undirected chain...

Study smarter with the SolutionInn App


