There are four resonance structures for anthracene, one of which is shown. Draw the otherthree. Anthracene
Question:
There are four resonance structures for anthracene, one of which is shown. Draw the otherthree.
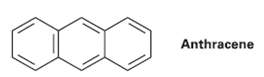
Transcribed Image Text:
Anthracene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
For your analysis, choose 10 countries, one of which is the United States. Create a table showing whether each country applies a worldwide or territorial approach to international income taxation....
-
For your analysis, choose 10 countries, one of which is the United States. Create a table showing whether each country applies a worldwide or territorial approach to international income taxation....
-
There are two contributing resonance structures for an anion called acetaldehyde enolate, whose condensed molecular formula is CH2CHO-. Draw the two resonance contributors and the resonance hybrid,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Recruiting people for jobs that require international assignments is increasingly important for many organizations. Where might an organization go to recruit people interested in such assignments?
-
Why has the number of U.S. banks operating overseas declined in recent years?
-
For each of the following sets of numbers, calculate a \(95 \%\) confidence interval for the mean ( \(\sigma\) known); before going through the steps in calculating the confidence interval, the...
-
Some of the account balances of Mali Company at December 31, 20x0 are shown below: 6% Preferred Stock ($100 par, 2,000 shares authorized) ... $ 20,000 PCIEP, Preferred .................. 3,000 Common...
-
3. (8 Points) Seven women and nine men are on the faculty in the mathematics department at a school. a. How many ways are there to select a committee of five members of the department if at least one...
-
It is September 2019 and you have just met with Ron Smith. Ron owns a battery-operated children's car manufacturing company, Car-It, and needs your advice on strategy. Ron is a self-made car builder....
-
Look at the three resonance structures of naphthalene shown in Section 15.7, and account for the fact that not all carboncarbon bonds have the same length. The C1C2 bond is 136 pm long, whereas the...
-
There are five resonance structures of Phenanthrene, one of which is shown. Draw the otherfour. Phenanthrene
-
Distinguish job-costing systems using sequential tracking from backflush costing.
-
How high is the image of the insect if the focal length of the lens is f = -125 mm? Follow the sign conventions. Express your answer to three significant figures and include the appropriate units....
-
If you wish to warm 44 kg of water by 10 C for your bath, find what the quantity of heat is needed. Express your answer in calories cal Express your answer to two significant figures and include the...
-
7. What is the tangential velocity associated with the revolution of Jupiter around the Sun? (distance from Jupiter to the Sun is 1,250,000,000 miles). revolve around the Sun. Assume that it takes...
-
A pipe discharges storm water into a creek. Water flows horizontally out of the pipe at 2.5 m/s, and the end of the pipe is 1.2 m above the creek. What is the speed of the water when it meets the...
-
Find f(g(x)) and g(f(x)) and determine whether the pair of functions f and g are inverses of each other. f(x)=x-2 and g(x)=x+2 f(g(x)) =
-
Write a program in \(\mathrm{R}\) that generates a sample \(X_{1}, \ldots, X_{n}\) from a specified distribution \(F\), computes the empirical distribution function of \(X_{1}, \ldots, X_{n}\),...
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
The equilibrium constant for the reaction A(g) B(g) is 10. A reaction mixture initially contains [A] = 1.1 M and [B] = 0.0 M. Which statement is true at equilibrium? (a) The reaction mixture...
-
Draw the complete structures of the following peptides: (a) Thr-Phe-Met (b) Serylarginylglycylphenylalanine (c) IMQDK (d) ELVIS
-
Draw the structure of the phenylthiohydantoin derivatives of (a) Alanine (b) Tryptophan (c) Lysine (d) Proline
-
Show the third and fourth steps in the sequencing of oxytocin.
-
Complete the table by: i. Defining each type of taxable transaction data provided below. ii. Listing one unique source of information for each type of taxable transaction data listed below. iii....
-
You are a partner in a CPA firm. You have a client that is being audited by the IRS. The IRS is using an indirect method of proof of his income. Your client is an attorney but not a tax attorney and...
-
Duncan Corp enters into a contract with Perry Company to sell four different products and each of the products is considered a separate performance obligation. The total price of the contract is...

Study smarter with the SolutionInn App


