Question: Use a computer-aided, steady-state simulation program to estimate the K-values, using the P-R and S-R-K equations of state, of an equimolar mixture of the two
Use a computer-aided, steady-state simulation program to estimate the K-values, using the P-R and S-R-K equations of state, of an equimolar mixture of the two butane isomers and the four butane isomers at 220?F and 276.5 psia. Compare these values with the following experimental results [J. Chem. Eng. Data, 7, 331(1962)]:
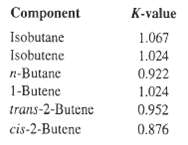
Component K-value Isobutane 1.067 Isobutene 1.024 n-Butane 0.922 1-Butene 1.024 trans-2-Butene 0.952 cis-2-Butene 0.876
Step by Step Solution
3.58 Rating (162 Votes )
There are 3 Steps involved in it
Using the ChemCAD process simulation program the foll... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (44).docx
120 KBs Word File


