Use heavy and dashed wedged hues to show the shapes of the following molecules. Show the bond
Question:
Use heavy and dashed wedged hues to show the shapes of the following molecules. Show the bond dipole of each polar bond and show the overall dipole of each molecule.
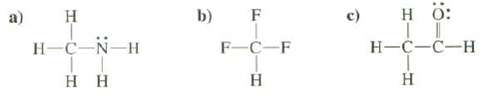
Transcribed Image Text:
a) H H-C-N-H HH b) F F-C-F H HÖ: T H-C-C-H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Use VSEPR theory to determine the geometry of the m...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the shapes of the following molecules a. CF 4 b. PCl 6 c. CO 2 d. Bh 3
-
Which of the following molecules have tetrahedral bond angles? H2O H3O+ +CH3 BF3 NH3 +NH4 -CH3
-
The ammonia molecule (NH3) has a dipole moment of 5.0 X 10-30 C m. Ammonia molecules in the gas phase are placed in a uniform electric field E with magnitude 1.6 X 106 N/c. (a) What is the change in...
-
The beam shown below is supported by a pin at A and a roller at B. The weight of the beam is 12 kN and a 15 kN force is applied 3 m to the right of A as shown. Determine the reaction forces at A and...
-
Explain why some organisations consider it important to measure customer retention and customer acquisition.
-
What are the strategic issues and considerations that Michel Bendayan should take into consideration when deciding how to enter the Japanese market? Conduct situational and SWOT analyses to identify...
-
Carbon dioxide at a temperature of \(0{ }^{\circ} \mathrm{C}\) and a pressure of \(600 \mathrm{kPa}\) (abs) flows through a horizontal 40-mm-diameter pipe with an average velocity of \(2 \mathrm{~m}...
-
Consider the unadjusted trial balance of Spateness, Inc., at December 31, 2016, and the related month-end adjustment data. Adjustment data at December 31, 2016: a. Accrued service revenue at December...
-
1. Write a paragraph about the "Fermi Paradox". 2. Define the Megawatt, Gigawatt and Terawatt units of power. Access the internet to determine the latest available figure of total global power...
-
A resident of Alberta has taxable income from employment of $170,000. This individual is considering three investments of equal risk and wishes to determine the after-tax income for each: a. $57,000...
-
Draw a Lewis structure for carbon monoxide (CO). Calculate the formal charges on the atoms and comment on the stability of this compound.
-
Predict the geometry at each atom, except hydrogen's, in these compounds: a) H --- 1 H b) H-c-c-c-
-
How largo a sample is required to be 95% confident that, at least 85% of the distribution of measurements is included between the sample extremes?
-
What is the National Practitioner Data Bank, and why was it created?
-
What is meant by empirical study in economics?
-
Describe the importance of employee relations and engagement efforts by HSOs, and give some examples of these activities.
-
Why do HSOs offer incentive compensation programs? How do these programs differ from base compensation programs?
-
Contrast employee assistance programs and employee suggestion programs.
-
If uranium were to split into 90 pieces of equal size instead of two, would more energy or less energy be released? Why? (a) less energy, because of less mass per nucleon (b) less energy, because of...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
Draw the graph of the function y = 1/1 x.
-
Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the C ? C1 bonds: CI C-H Chloromethane H.
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
Make three-dimensional drawings of the following molecules, and predict whether each has a dipole moment. If you expect a dipole moment, show its direction. (a) H2C = CH2 (b) CHC13 (c) CH2C12 (d) H2C...
-
Suppose you were interested in studying the quality of conditions within a prison. What indicators would you measure to give the clearest picture of the realities of prison life? Cite the below...
-
On March 31, 2023, Panda Co. assessed its assets for impairment as part of its year-end procedures. It was found that equipment had a recoverable value of $15,000, a remaining useful life of three...
-
Petty's comparative balance sheets at December 31, 2020, and December 31, 2019, report the following (in millions). (Click the icon to view the comparative balance sheets.) Requirements Below are...

Study smarter with the SolutionInn App


