Question: Use the density-of-states function, Equation 27-28, to estimate the fraction of the conduction electrons in copper that can absorb energy from collisions with the vibrating
Use the density-of-states function, Equation 27-28, to estimate the fraction of the conduction electrons in copper that can absorb energy from collisions with the vibrating lattice ions at
(a) 77 K and
(b) 300K.
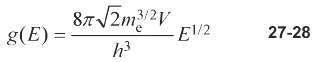
87 /2m 2V g(E) 27-28 E/2 h3
Step by Step Solution
3.51 Rating (178 Votes )
There are 3 Steps involved in it
a Using the result of Problem 34 ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
10-P-E-M-E-C (29).docx
120 KBs Word File


