Question: Using a curved fishhook arrow, propose a mechanism for formation of the cyclopentane ring of prostaglandin H2. What kind of reaction isoccurring? CO2H
Using a curved fishhook arrow, propose a mechanism for formation of the cyclopentane ring of prostaglandin H2. What kind of reaction isoccurring?
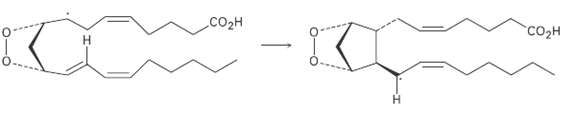
CO2H "
Step by Step Solution
3.29 Rating (161 Votes )
There are 3 Steps involved in it
Strategy The tails of the arrows show the location of ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-O-C (165).docx
120 KBs Word File


