Vanillin (the natural vanilla flavoring) occurs in nature as a -glycoside of glucose. Suggest a structure for
Question:
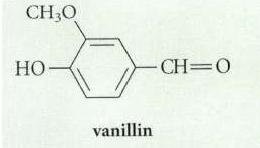
Transcribed Image Text:
CH,O vanillin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Because naturally occurring glycosides generally ha...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Vanillin is the active component of natural vanilla flavoring. OCH vanillin
-
Pectin, which occurs in plant cell walls, exists in nature as a polymer of D-galacturonic acid methylated at carbon 6 of the monomer. Draw a Haworth projection for a repeating disaccharide unit of...
-
Vanillin, the dominant flavoring in vanilla, contains C, H, and O. When 1.05 g of this substance is completely combusted, 2.43 g of CO2 and 0.50 g of H2O are produced. What is the empirical formula...
-
The adjusted trial balance for Ray Corporation at July 31, 2017, the corporation's fiscal year end, contained the following: Of the lease liability amount, $16,250 is due within the next year. Total...
-
Esther (All) God is not mentioned in this book. How does Gods providence show up in this book?
-
Consider a Carnot-cycle heat pump having 1 kg of nitrogen gas in a cylinder/piston arrangement. This heat pump operates between reservoirs at 300 K and 400 K. At the beginning of the low-temperature...
-
For pumping mustard oil from a drum, a suitable pump is (a) reciprocating pump (b) vane pump (c) centrifugal pump (d) gear pump
-
A turbine discharges 200 kg/h of saturated steam at 10.0bar absolute. It is desired to generate steam at 250C and 10.0 bar by mixing the turbine discharge with a second stream of superheated steam of...
-
1. What is organization theories in action? 2. What is nokia organization theory in action?
-
Box and Liu (1999) describe an experiment flying paper helicopters where the objective is to maximize flight time. They used the central composite design shown in Table 14E.9. Each run involved a...
-
Into what other aldose and 2-ketose would each of the following aldoses be transformed on treatment with base? Give the structure and name of the alssose, and the structure of the 2-ketose. D-allose
-
Draw structures for: Is opropy 1-D-galactopyranoside
-
Explain the different classifications of principals and how the estition affects the liability of the principal for the actions of an agent.
-
1. Calculate the gravitational force FG on a mass m in relation to the distance x from point A (see figure) along the central line (dashed line): a) A b) 7 A a) inside and outside of a uniform,...
-
Book keeping is not a part of accounting. Do you agree with this statement? Give the rules of debit and credit? Write five limitations of ratio analysis? If net working capital for current year is...
-
How do organizations balance the needs and preferences of their employees with the constraints and goals of the organization when designing a compensation and benefits program?
-
Find the magnitude of the resultant force (FR) and the angle (p) it makes measured counterclockwise from the positive X-axis, given: FA= 50 lbs, 0A = 56, FB = 24 lbs, B = 28 FR = Number = Number lbs O
-
How do legal considerations, such as equal pay and minimum wage laws, affect the design of a compensation and benefits program?
-
Now write down what you might dislike about each form of business. For example, would you dislike the risk of bearing all legal responsibility in a sole proprietorship? Would you dislike having to...
-
Using the parallel-axis theorem, determine the product of inertia of the area shown with respect to the centroidal x and y axes. 6 in. 9 in. 9 in- 4.5 in. in. 4.5 in.
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
Make three-dimensional drawings of the following molecules, and predict whether each has a dipole moment. If you expect a dipole moment, show its direction. (a) H2C = CH2 (b) CHC13 (c) CH2C12 (d) H2C...
-
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane :O:
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


