What alkenes might be used to prepare the following alcohols by hydroboration/oxidation? (a) CH3 (b)
Question:
What alkenes might be used to prepare the following alcohols by hydroboration/oxidation?
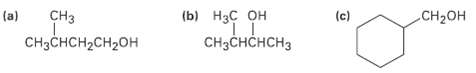
Transcribed Image Text:
(a) CH3 (b) НаС он CHзснснсHз (c) .CH-он CHзCHCH2CH2OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
Strategy As described in Worked Example 72 the strategy in this sort ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What variables might be used to segment these industrial markets? (a) Industrial sweepers, (b) Photocopiers, (c) Computerized production control systems, (d) Car rental agencies.
-
What approaches might be used to recognize risk in capital budgeting?
-
What arguments might be used to persuade shareholders in an acquisition target to reject offers at any price? Are they plausible?
-
Draw isometric projection of the following figure 10 40 50 $30 Pg. 3 : 20
-
Consider the following Excel regression of perceived sound quality as a function of price for 27 stereo speakers. (a) Is the coefficient of Price significantly different from zero at = .05? (b) What...
-
What is DeepFace and how is it used?
-
The following information was reported in the December 31, 2007, financial statements of Southwest Airlines, Inc. (listed alphabetically, amounts in millions). Accounts Payable $1,731 Accounts...
-
Effects of Burger Kings Current Liabilities on Its Statement of Cash Flows The following items are classified as current liabilities on Burger King Holdings, Inc.s balance sheets as of June 30, 2008,...
-
Calculate the predetermined over head rate assuring sandhill company estimates total manufacturing overhead costs of 1 0 0 8 0 0 0.
-
Ever since the day she took her first economics class in high school, Lydia wondered about the financial practices of her parents. They worked very hard to earn enough money to live a comfortable...
-
Show the structures of the products you would obtain by hydroboration/oxidation of the following alkenes: H (a) "CH (a) CH3C=CHCH2CH3 (b)
-
The following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structures of both, and explain the result.
-
Siren Company builds custom fishing lures for sporting goods stores. In its first year of operations, 2025, the company incurred the following costs. Siren Company sells the fishing lures for $25....
-
During the weeks preceding "Black Friday", many retail stores reduce their prices. What is the factor affecting suppliers and consumers before Black Friday?
-
The Year 5 general fund budget for Blake County included $ 5 0 , 0 0 0 budgeted for new uniforms. At Dec. 1 , Year 5 , the following account balances pertain to fire personnel uniforms:...
-
Show and discuss using a diagram how overproduction results in deadweight loss. Discuss how this issue can be addressed. Question 2 Discuss how changing input prices will impact the slope and...
-
Sales $ 1 5 9 , 0 0 0 , Variable costs $ 8 3 , 0 0 0 , Contribution margin $ 7 6 , 0 0 0 , fixed costs $ 9 1 , 0 0 0 and operating income ( loss ) $ - 1 5 , 0 0 0 , if it drops off the product line,...
-
A friend of mine just told me, "The fact that cross-price elasticity of demand for sport shoes and socks is negative means that people often buy sport shoes and socks together. Is my friend correct?...
-
The characteristic equation of a sampled system is \[z^{2}+(K-1.5) z+0.5=0\] Find the range of \(K\) so that the system is stable. (Answer: \(0
-
If the cylinder described in Problem 21.3 were initially heated to 500F, how long would it take for the center of the cylinder to cool to 240F if it were constructed of a. Copper? b. Brass? c. Nickel?
-
What is the dipoledipole force? How can you predict the presence of dipoledipole forces in a compound?
-
Rank the compounds within each of the sets shown in Fig. P7 .62 according to their heats of formation, loweet first. Explain. Fig. P7 .62 (a) (b) , H CH3 CH3 H CH
-
(a) What two diastereomeric products could be formed in the hydroboration-oxidation of the following alkene? (b) Considering the effect of the methyl group on the approach of the borane-THF reagent...
-
The ÎG° for the equilibrium in Fig. P7.67ais 4.73 kJ mol-1 (t.13 kcal mol-1. (The equilibrium favors conformation A.) (a) Which behaves as if it is larger, methyl or phenyl (Ph)? iWhy is...
-
Discuss the main distinguishing features of majority, concurring, and dissenting opinions
-
On September 3, 2023, Wilkins purchased 1244 stock in Brass Corporation for $6,000. On December 31, 2023, the stock was worth $8,500. On August 15, 2024, Wilkins was notified that a. 2023-$0;...
-
Kowalczyk Company spends $30000 dollars for manufacturing equipment. When should the cost of the equipment be expensed? Over the period that the equipment will be used When the performance obligation...

Study smarter with the SolutionInn App


