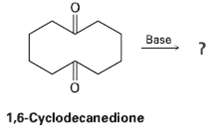
What product would expect to obtain from base treatment of 1,6-cyclo-decacedione? Base 1,6-Cyclodecanedione
Question:
What product would expect to obtain from base treatment of 1,6-cyclo-decacedione?
Transcribed Image Text:
Base 1,6-Cyclodecanedione
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
This intramolecular aldol con...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What product would you expect to obtain from the base-catalyzed Michael reaction of (a) 1, 3-diphenylprop-2-en-1-one (Section 19.5A) and acetophenone (b) 1, 3-diphenylprop-2-en-1-one and...
-
What product would you expect to obtain from each of the following reactions? a. b. c. CH3CH2CHCH CH2CH2COH CH COCH CH HCI CH OH CH CH COH 1. SOC2 2. AICI3 3. H20
-
What product would you expect to obtain from each of the following reactions? (a) (b) NaNO2, HCI SO2Cl aq. KOH
-
Bilge Pumpworks and Seaworthy Rope Company agreed to merge on January 1, 20X3. On the date of the merger agreement, the companies reported the following data: Bilge Pumpworks has 10,000 shares of its...
-
Major professional schools are highly competitive and most applicants do not get past the admissions process. That makes admissions a critical gatekeeper function for these schools. Given that, why...
-
Consider Exercise 2.58 on page 79. Can it be said that the ratings given by the two experts are independent? Explain why or why not. Exercise 2.58 Two tire-quality experts examine stacks of tires and...
-
Describe how wrongful birth, wrongful life, and wrongful conception differ.
-
Sams Company reported the following stockholders equity account balances on December 31, 2011. Preferred stock (12%, $100 par value, call price is $105)... $100,000 Common stock, $10 par...
-
How can appreciative inquiry methodologies be integrated into change leadership practices to catalyze positive organizational change by leveraging existing strengths and fostering a culture of...
-
Jerrys Jellies sells one-and two-year mail-order subscriptions for its jelly-of-the-month business. Subscriptions are collected in advance. An analysis of the recorded sales activity revealed the...
-
Treatments of a 1, 3-diketone such as 2,4-pentanedione with base does not give an aldol condensation product. Explain.
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
What is the relationship between windows and frames?
-
Joan is a 17-year-old refugee who arrived in Australia as an unaccompanied minor four (4) years ago. She has been granted permanent residency status. She has difficulty with written English and has...
-
determine the future value of a present value of $37,500 at 2% annual interest compounded monthly for 17 years, and determine the amount of interest earned. The future value is and the interest...
-
Which strategy is an appropriate way for teachers to let classroom volunteers know they are appreciated? explain
-
why are ethnographic methods such as participant-observation useful for doing research on food and culture?
-
An investment company considers to purchase of the Apartment Lakeshore project. The year 1 NOI is expected to be $2,000,000, and based on the recent economic forecast, market supply and demand and...
-
Walter Corporation reports $500,000 of taxable income in the current year. Determine its book income if tax depreciation was $15,000 more than book depreciation, its deductible meals and...
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
Diazomethane is a highly poisonous, explosive compound because it readily evolves N 2 . Diazomethane has the following composition by mass: 28.57% C; 4.80% H; and 66.64% N. The molar mass of...
-
Give the structures of the following compounds. (a) Methylene iodide (b) Carbon tetrabromide (c) 3-bromo-2-methylpentane (d) Iodoform (e) 2-bromo-3-ethyl-2-methylhexane (f) Isobutyl bromide (g)...
-
Draw a perspective structure or a Fischer projection for the products of the following SN2 reactions. (a) trans-1-bromo-3-methylcyclopentane + KOH (b) (R)-2-bromopentane + KCN (c) (d) (e) (f) CH Br-H...
-
Under appropriate conditions, (S)-1-bromo-1-fluoroethane reacts with sodium methoxide to give pure (S)-1-fluoro-1-methoxyethane. (a) Why is bromide rather than fluoride replaced? (b) Draw perspective...
-
Silverton Confectionery is a growing Berkshire-based company specialising in selling quality chocolates and sweets at higher than average prices through newsagents and confectioners. At present their...
-
4. X, the proprietor of a departmental store, decided to calculate separate profits for his two departments L and M for the month ending 31st January. Stock on 31st January could not be valued for...
-
What level of confidentiality should be attached to the preparation and handling of a memorandum of law? Why? Assume you have been working for a legal specialist in estate law for a number of years...

Study smarter with the SolutionInn App


