What products would you expect from reaction of the following alkenes with NBS? If more than one
Question:
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall.
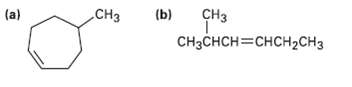
Transcribed Image Text:
сHз (a) .СНз (b) CнзснсH3Dснсн2СHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a CH3 5Methyl cycloheptene NBS CC14 heat CH3 Br 3Bromo5methyl cycloheptene Br C...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from reaction of 1-methylcyclohexene with the following reagents? (a) Aqueous acidic KMnO4 (b) O3, followed by Zn, CH3CO2H
-
What products would you expect from each of the following reactions? (a) (b) (c) t-BuOK CHBr CH2Zn(Cu) diethyl ether
-
What product would you expect from reaction of 1 equivalent of methanol with a cyclic anhydride, such as phthalic anhydride (1, 2-benzenedicarboxylic anhydride)? What is the fate of the second ?half?...
-
A saving bond earns a variable rate of interest that can change six months, with compounding done monthly. The initial rate was 6.8% in early 2015. If that rate continues unchanged for the 3 years of...
-
Consider the stylized example in Figure 7.13. Repeat the forecasts using the additive model with trend, using the same smoothing constants. FIGURE 7.13 Stylized Model with Trend and Cyclic...
-
The following data were extracted from the income statement of Keever Inc.: a. Determine for each year (1) The inventory turnover and (2) The number of days sales in inventory. Round to the nearest...
-
A printed circuit board has eight different locations in which a component can be placed. If four different components are to be placed on the board, how many different designs are possible?
-
Sylvia Manufacturing Inc. had sales of $1,800,000 for the first quarter of 2012. In making the sales, the company incurred the following costs and expenses. Prepare a CVP income statement for the...
-
Fairfield Company's raw materials inventory transactions for the most recent month are summarized here: Note: Assume, purchase of raw materials is on account. Beginning raw materials Purchases of raw...
-
A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term government and corporate bond fund, and the third is a T-bill money market fund that...
-
The major product of the reaction of methylenecyclohexane with N-bromo-succinimide is 1-(bromomethyl) cyclohexane. Explain CH2Br CH2 NBS Major product
-
How would you prepare the following alkyl halides from the correspondingalcohols? CH (a) CI (b) Br CH CHH2CH CH (c) CI H (d) BrCH2CH2CH2CHCH CHCH2H2cH3 CH
-
The Wellton Fund is a balanced mutual fund that includes a mix of stocks and bonds. Following are the year-end share prices of the fund and Dow Jones Industrial Average (DJIA) for a 20-year period:...
-
import javax.swing.JOptionPane; public class GUI_DropDownList { public static void main(String[] args) { int answer=0; int x=JOptionPane.showConfirmDialog(null,"Do you want to continue...", "OPTION",...
-
Avery, age 42, is unmarried and has two qualifying dependents for whom she maintains a household for the entire year. Avery has an adjusted gross income of $75,000 and uses the standard deduction....
-
Using the AICPA Code of Professional Conduct Links to an external site. describe how this was either applied, misapplied, or completely ignored. Using the COSO Internal ControlIntegrated Framework...
-
Company A has the following data: Prime costs are 180% of direct materials costs. DM costs for year 20xx are $ 100,000. The company had estimated the overhead costs to be $400,000 and the estimated...
-
suppose your company shipped product on december 12. it was reveived by the customers and placed in inventory on december 31,2024. but not signed off on until january 2, 2025.does SAB 101 say that...
-
The British Broadcasting Corporation (BBC) conducts an annual "Price of Football" study. The 2013 version suggested that an \(11 \%\) increase in the price of football tickets resulted in a drop of...
-
How is use of the word consistent helpful in fraud reports?
-
Which metal is most easily oxidized? (a) Na (b) Cr (c) Au
-
The heat of combustion of cis-1, 2-dimethylcyclopropane is larger than that of the trans isomer. Which isomer is more stable? Use drawings to explain this difference in stability.
-
Trans-1, 2-Dimethylcyclobutane is more stable than cis-1, 2-dimethylcyclobutane, but cis-1, 3-dimethylcyclobutane is more stable than trans-1, 3-dimethylcyclobutane. Use drawings to explain these...
-
Name the following alkanes and haloalkanes. When two or more substituents are present, list them in alphabetical order. (a) (b) (c) (d) CH CH CH-CH CHCH Br CH CH CH CH CH CH CH CH, CH,CH(CH3)....
-
Which routing criteria is most commonly used? What units does it use?
-
Alhamra Technology's capital structure is as follows [in %]: Debt 30; Preferred stock 20; Common equity 50; The before tax cost of debt is 8 percent and tax rate is 50%; the cost of preferred stock...
-
Cohen and Brawer (2003) express that there are over 300,000 faculty nationwide in the community college system and over 180,000 are part time. This means that about two thirds of the community...

Study smarter with the SolutionInn App


