What products would you expect from the followingreactions? ta) CH3CH2CH2C=CH + 2 Cl2 () (b) 1 HBr
Question:
What products would you expect from the followingreactions?
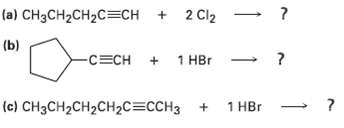
Transcribed Image Text:
ta) CH3CH2CH2C=CH + 2 Cl2 (ь) (b) 1 HBr -СЕCH + 1 нBr -C=CH c) CH3CH2CH2CH2C3СCHЗ + 1 HBr - ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Markovnikov addition is observed with alkynes as ...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from the reaction of ethylmagnesium bromide (CH3 CH2MgBr) with each of the following reagents? (a) H2O (b) D2O (c) (d) (e) (f) (g) then H O "H Ph" PhPh, then NH4CI, HO...
-
What products would you expect from the following reactions? (a) (b) (c) (d) (e) OMe OMe
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
Which subject does Corporate social responsibility belong to?
-
A firm is trying to decide whether to enter a highly uncertain market now or to wait to decide two years from now, when the size of the market will be less uncertain. If it enters now it must invest...
-
Why would a network manager benefit from having network management tools? Describe five scenarios.
-
Phone Screens and Computer Screens are two divisions operated as investment centres of Malta Ltd. Management wants to know which of the two earned the highest return on investment for the year ended...
-
An examiners close inspection of the annual financial statements and the accounting records revealed that Mawani Inc. may have violated some accounting principles. The examiner questioned the...
-
At which value(s) for x does the graph of y = x-3x - 10x + 7 have horizontal tangent lines?
-
Consider the following simple offshoring model of the type described in Section 11.2. The United States and Mexico both produce radios, using skilled and unskilled labor. Each radio requires three...
-
There are seven isomeric alkynes with the formula C6H10. Draw and name them.
-
What product would you obtain by hydration of the following alkynes? (a) CH3CH2CH2C=CCH2CH2CH3 CH (b) CCH2CECH-CH2CH3
-
What is a cost-benefit analysis?
-
Question 5: Consider the exchange economy of Nirvana, which consists of two people, A and B. Two goods are available for consumption, wine and chocolate. The endowment of the economy is 64 units of...
-
A firm's profit is related to the number of sales persons (S) and the price of the product (P) according to the equation: = 150S.2p-1.4 A. Use partial differentials to calculate the percentage change...
-
On January 1, 2024, the Mason Manufacturing Company began construction of a building to be used as Its office headquarters. The building was completed on September 30, 2025. Expenditures on the...
-
You manage a burger restaurant on a busy corner in the corporate district of town. Across the street is your competitor, who also makes burgers. Business is good for both burger restaurants, and at...
-
Consider the market for software engineers. Suppose there is a decrease in labor demand. We assume the following: . It takes time to produce a software engineer. Agents decide to become software...
-
Exactly 380 girls. Assume that 400 births are randomly selected. Use subjective judgment to determine whether the given outcome is unlikely, and also determine whether it is unusual in the sense that...
-
Design a circuit which negative the content of any register and store it in the same register.
-
Complete and balance each gas-evolution equation. a. HBr(aq) + NIS(s) b. NH4l (aq) + NaOH(aq) c. HBr(aq) + NaS(aq) d. HCIO4(aq) + LiCO3(aq)
-
The artificial sweetener aspartame (sidebar, p. 1208) was withheld from the market for several years because, on storage for extended periods of time in aqueous solution, it forms a diketopiperazine...
-
Identify each of the compounds A-D in the reaction scheme shown in Fig. P26.67, p. 1331. Explain your answers. H,N NH NaNO /HCI CH(CH3)2 HCL H20 C -heat (CHs)-CH-CH-=O + D
-
When peptides containing the Asn-Gly sequence, such as H in the equation given in Fig. P26.69, are stored in aqueous solution at neutral or slightly basic solution, ammonia is liberated and a...
-
State the dividend irrelevance proposition. What are the assumptions behind this proposition? Explain why this proposition does not hold in the real world. (20 marks)
-
How many monthly withdrawals of $1,400 will an investment of $75,000 sustain if the first withdrawal is made 12 months from now and the money earns 8.4% compounded monthly
-
1. You buy a bond with 3 years left to maturity and a yield to maturity of 6% for $920. After 1 year you receive a coupon payment of $30 and sell the bond for $940. What was your rate of return on...

Study smarter with the SolutionInn App


