What product(s) would you expect from the reaction of 1-methylcyclohexene with NBS? Would you use this reaction
Question:
What product(s) would you expect from the reaction of 1-methylcyclohexene with NBS? Would you use this reaction as part of asynthesis?
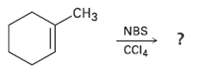
Transcribed Image Text:
CHз NBS CCI4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Three different allylic radical intermediates can be ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What product(s) would you expect from the reaction of 1, 4-hexadiene with NBS? What is the structure of the most stable radical intermediate?
-
What products would you expect from the reaction of 1-bromopropane with each of the following? (a) NaNH2 (b) KOC (CH3)3 (c) NaI (d) NaCN (e) NaC CH (f) Mg, then H2O
-
What products would you expect from the following reactions? (a) (b) PdC PPh3. Et N Br Br Pd(OAc)h PPhy. EtyN
-
There are different types of non-probability sampling methods. Below are examples of such methods. You are required to go through them and (for each example); a. Name the Sample Method b. Write a not...
-
The database Population.xlsx contains data on the populations of the 50 states from 1990 to 1999. a. How many records are in this database? b. How many fields are in this database? c. Are there any...
-
Consider the fourth-order plus time delay system represented below: Assume that a step change is applied in the system input U(s). Will the time required for the output Y(s) to reach steady state...
-
Should a board member have personal liability insurance coverage?
-
Barreras Outdoor Outfitters sells many items that sporting enthusiasts find useful. The company sells shoes, pants, shirts, jackets, fly-fishing equipment, hiking equipment, hunting equipment, and...
-
Let's suppose you had two potential segments to consider for targeting. Segment A is of moderate market attractiveness and your firm has an advantageous competitive position. Segment B has a high...
-
What is the name of the court of general jurisdiction in your state? What is the name of a limited-jurisdiction court in your state? What is the subject matter jurisdiction of this court? This...
-
A chemist requires a large amount of 1-bromo-2-pentene as starting material for a synthesis and decides to carry out an NBS allylic bromination reaction. What is wrong with the following synthesis...
-
How would you prepare the following compounds, starting with cyclopentane and any other reagents needed? (a) Chloro cyclopentane (b) Methylcyclopentane (c) 3-Bromocyclopentene (d) Cyclopentanol (e)...
-
NET The Internet is filled with Webcams. For example, Pebble Beach Golf Club has several Webcams (www .pebblebeach.com). How could a researcher use Webcams like these to collect behavioral data?
-
A challenge analysis matrix, what does an area with low performance and high importance indicate?
-
Analyze the historical events or circumstances that created a need for the legislative act you have selected.
-
1. Define option. What does "exercising the option" mean? What is the strike or exercise price? What is the expiration date for an option and what is true of the option after the expiration date? How...
-
2. Consider the following position: Buy the Home Depot Nov 125 call @ $1.98 and write the Home Depot Nov 125 put @ $2.98. Shares of Home Depot are currently selling at $123.83. a) Find the cost of...
-
MARR is 12% . Initial investment is 30000. Annual savings is 13000. Years is 7. what is the annual worth
-
The concentration of the active ingredient, sodium hypochlorite, in Clorox is twice that of the generic brand. Consequently, Chris views one cup of Clorox to be a perfect substitute for two cups of...
-
Give an example of transitory income. What effect does this income have on the marginal propensity to consume?
-
The titration of 15.00 mL of HBr solution of unknown concentration requires 18.44 mL of a 0.100 M KOH solution to reach the equivalence point. What is the concentration of the unknown HBr solution in...
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
Rank the following carbocations in decreasing order of stability. Classify each as primary, secondary, or tertiary. (a) The isopentyl cation, (b) The 3-methyl-2-butyl cation, (c) The 2-methyl-2-butyl...
-
Each of the following proposed mechanisms for the free-radical chlorination of methane is wrong. Explain how the experimental evidence disproves each mechanism. (a) Cl2 + hv Cl2* (an activated form...
-
What are some of the best ways for attaining skills in active listening?
-
. Do you agree with the following statement: "English is a powerful tool that has become entrenched in business processes worldwide-whether for analyzing stocks or issuers, budgeting, or organizing...
-
As a roofer business owner describe the importance of effective communication in your current work/role?

Study smarter with the SolutionInn App


