When a primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an
Question:
When a primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out at higher temperature, an alkyl chloride is often formed.Explain.
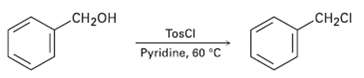
Transcribed Image Text:
CH2CI CH2он TosCI Pyridine, 60 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
Tos you Pyr CH0H CI CHOTOS CHCl OTOS PyrH At lower temperatures a tosylat...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A carbohydrate (S) decomposes in the presence of an enzyme (E) The Michaelis-Menten kinetic parameters were found to be as follows M 200 mol m 3 100 mol m 3 min a Calculate the change of substrate...
-
Alkyl halides add to alkenes in the presence of AlCl3; yields are the highest when tertiary halides are used. Predict the outcome of the reaction of tert-pentyl chloride (1-chloro-2,...
-
When benzene is treated with I 2 in the presence of C u Cl 2 , iodination of the ring is achieved with modest yields. It is believed that C u Cl 2 interacts with I 2 to generate I + , which is an...
-
Rainbow manufactures wooden backyard playground equipment. Rainbow estimated $1,785,000 of manufacturing overhead and $2,100,000 of direct labour cost for the year. After the year was over, the...
-
What challenges do current college graduates face?
-
Should one society force its work standards and wage rates on another? Why or why not?
-
A supplier of imported vernier calipers claims that \(90 \%\) of their instruments have a precision of 0.999. Testing the null hypothesis \(p=0.90\) against the alternative hypothesis \(p eq 0.90\),...
-
Hedley Company has received a special order for Product R3P at a selling price of $20 per unit. This order is over and above normal production, and budgeted production and sales targets for the year...
-
Write a script that does the following: Defines a variable, N = 1000 Generates a vector of N random samples from a normal (Gaussian) distribution with a mean of 100 and standard deviation of 10....
-
Roadrunner Bookstore uses the perpetual inventory system. Roadrunner Bookstore is owned by Tina Smith. The company had the following transactions for the month of September. Sep 1 Sep 2 Sep 4 Sep 5...
-
Compound X is optically inactive and has the formula C16H16Br2. On treatment with strong base, X gives hydrocarbon Y, C16H14. Compound Y absorbs 2 equivalents of hydrogen when reduced over a...
-
SN2 reactions take place with inversion of configuration, and SN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of...
-
Given a list of web pages, create a symbol table of words contained in those web pages. Associate with each word a list of web pages in which that word appears. Write a program that reads in a list...
-
9 years ago Clarissa purchased shares in a certain mutual fund at Net Asset Value (NAV) of $52. She reinvested her dividends into the fund, and today she has 7.9% more shares than when she started....
-
The true root of an equation is 1 . 0 1 . One student chooses an initial interval of [ 1 , 2 ] to start the bisection method. What is the absolute true percentage error after 4 steps?
-
Lynne needs to borrow $8500 for cosmetic surgery. She obtains a loan from her grandmother for 54 months at a simple interest rate of 7.6%. What is the loans future value?
-
A new bond issue has an annual yield of 5.6%. The bond pays coupons twice a year . What is the effective annual yield (EAY) on the investment?
-
You have been searching the title of a particular parcel of property as part of the preclosing procedures and notice that a deed recorded in 1977 indicates in the encumbrance clause that the property...
-
CustomMetalworks in Oregon produces guy wire attachments for towers (cell, broadcasting, etc.) according to customer order. The company has determined that guy attachments can be produced on three...
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
What is the definition of the reaction quotient (Q) for a reaction? What does Q measure?
-
Write equations showing the expected products of the following enamine alkylation and acylation reactions. Then give the final products expected after hydrolysis of the iminium salts. (a) Pyrrolidine...
-
(A true story.) Chemistry lab students added an excess of ethylmagnesium bromide to methyl furoate, expecting the Grignard reagent to add twice and form the tertiary alcohol. After water workup, they...
-
Propose a mechanism for the following reaction. Show the structure of the compound that results from hydrolysis and decarboxylation of the product. CHO + CH2(COOCH2CH3)2 hydrolysis, decarboxylation...
-
This number is the "federal adjusted gross income" according to the line title. What do we call this process of starting our state return with a number from the federal return?
-
What is the main reason for a franchisee to have a contingency fund?
-
Kalan purchased four new tires at Walmart for $1,000 using there "no payments for 12 months plan." About a year later, before Kalan had made any payments, the tires were not performing as well as...

Study smarter with the SolutionInn App


