Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist? a) CHCHCH=CHCH CH3
Question:
Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist?
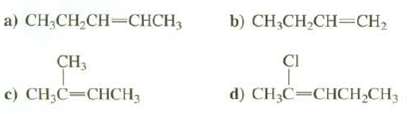
Transcribed Image Text:
a) CH₂CH₂CH=CHCH₂ CH3 c) CH₂C=CHCH₂ b) CH3CH₂CH=CH₂ CI d) CH₂C=CHCH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
Cistrans isomers are stereo isomers that differ in the placement of groups about ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Which of these compounds can be a member of an isomer pair? In each case where isomerism is possible, identify the type or types of isomerism. [Sections 24.2, 24.4] CH2 C C-OH --O NHE Cl (b) CH3CH2CH...
-
Draw the cis and trans isomers for the following compounds: a. 1-ethyl-3-methylcyclobutane b. 2-methyl-3-heptene c. 1-bromo-4-chlorocyclohexane d. 1,3-dibromocyclobutane
-
What Codification citation indicates how unrealized gains or losses from changes in fair value affect net income when investments are classified as available-for-sale?
-
Distinguish between a support department and a production department. Give an example of a production department in a bank, and a support department in a restaurant chain.
-
The price of a stock has gone up from $24 to $35 in one year. It also paid a year-end dividend of $1.20. What is the stock's (a) Income yield, (b) Capital gains yield, (c) Total return?
-
Describe the concept of the life cycle object, how it relates to collection objects, and give an example of a life cycle class with appropriate attributes.
-
Data for investments in stock classified as trading securities are presented in E16-10. Assume instead that the investments are classified as available-for-sale securities. They have the same cost...
-
You purchase 100 shares of stock for $50 a share. The stock pays a $4 per share dividend at year-end. a. What is the rate of return on your Investment If the end-of-year stock price Is (1) $46; (II)...
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
Explain which compound has the higher solubility in water.
-
Draw the cis-trans isomers for these compounds and explain which is more stable: CH3 a) CH CH CHCOH NH c) e CH, CH2NHCH CH f CH3CH CH CH
-
a. Prepare a context diagram and at least two levels of DFDs for this process. Level 0 Data Flow Diagram for a course registration system: Level 1 Data Flow Diagram for a registration system: b....
-
a. Show that \[s_{y}^{2}=\frac{1}{n-1} \sum_{i=1}^{n}\left(y_{i}-\bar{y} ight)^{2}=\frac{1}{n-1}\left(\sum_{i=1}^{n} y_{i}^{2}-n \bar{y}^{2} ight) .\] b. Follow the same steps to show...
-
A box of volume \(V\) has a movable partition separating it into two compartments. The left compartment contains 3000 particles, the right one contains 1000 particles, and initially the partition is...
-
A cryogenic substance is found to have a specific heat capacity \(c\) that varies with temperature according to \(c=\beta T^{2}\), where \(\beta\) is an empirically derived constant with units...
-
According to the Can Manufacturers Institute, the energy used to make an aluminum can from recycled aluminum is \(5 \%\) of the energy used to make an aluminum can from virgin ore. In a typical year,...
-
To make yourself some coffee, you put one cup of water \((236 \mathrm{~g})\) in a small pot on the stove. What quantity of energy must be transferred thermally to the water to raise its temperature...
-
Piper High School is in Piper, Kansas, a town located about 20 miles west of Kansas City, Missouri. Christine Pelton was a high school science teacher there. Ms. Pelton, age 26, had a degree in...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
The region bounded by the curves y = arctan x, y = 0, and x = 1 is rotated about the y-axis. Find the volume of the resulting solid.
-
(a) How many degrees of unsaturation does a-terpinene have? (b) How many double bonds and how many rings does it have? (c) Propose a structure fora-terpinene. CCH-CH2CCHCH3 C-C H ...
-
Evidence that cleavage of 1, 2-diols by HIO 4 occurs through a five-membered cyclic periodate intermediate is based on kinetic data?the measurement of reaction rates. When diols A and B were prepared...
-
Reaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis- and trans-1-bromo-3-methylcyclohexane and cis- and trans-1-bromo- 2-methylcyclohexane. The analogous reaction of HBr...
-
Discuss the main distinguishing features of majority, concurring, and dissenting opinions
-
On September 3, 2023, Wilkins purchased 1244 stock in Brass Corporation for $6,000. On December 31, 2023, the stock was worth $8,500. On August 15, 2024, Wilkins was notified that a. 2023-$0;...
-
Kowalczyk Company spends $30000 dollars for manufacturing equipment. When should the cost of the equipment be expensed? Over the period that the equipment will be used When the performance obligation...

Study smarter with the SolutionInn App


