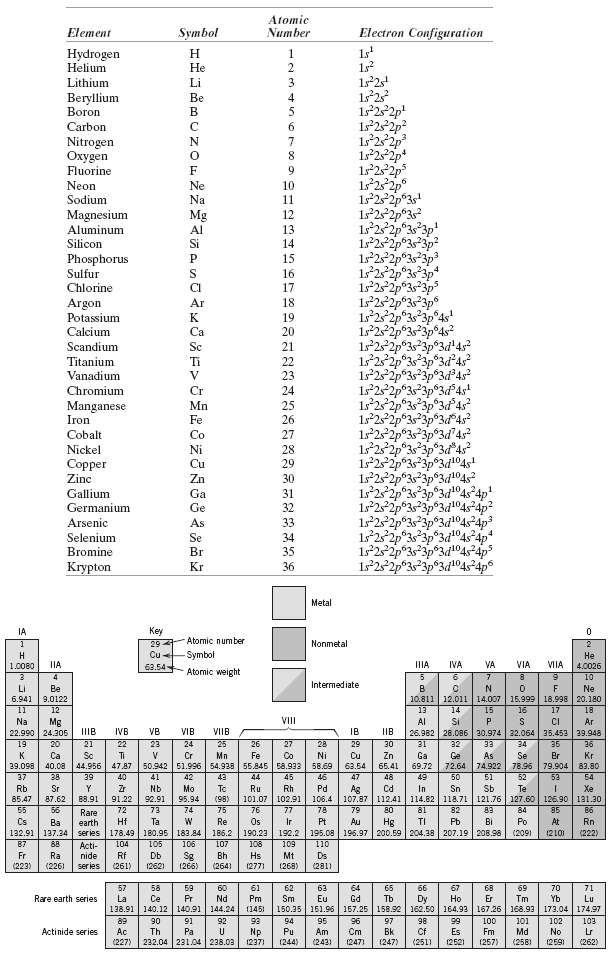
Question: Without consulting figure?or Table 2.2, determine whether each of the electron configurations given below is an inert gas, a halogen, an alkali metal, an alkaline
Without consulting figure?or Table 2.2, determine whether each of the electron configurations given below is an inert gas, a halogen, an alkali metal, an alkaline earth metal, or a transition metal. Justify your choices.
(a) 1s22s22p63s23p63d74s2
(b) 1s22s22p63s23p6
(c) 1s22s22p5
(d) 1s22s22p63s2
(e) 1s22s22p63s23p63d24s2
(f) 1s22s22p63s23p64s1
Atomic Number Electron Configuration Element Symbol 1s' 12 Hydrogen Helium 2 Lithium Li 122 1s2s2p! 1s 22p? 1s 22p 1s 2-2p 12 2p 122p 1s2s2p3s 1s25 2p3 Is22p3s 3p 1s 2 2p 1s 25 2p Beryllium Boron Be 4 Carbon 6. Nitrogen en Fluorine Neon Sodium Ne 10 Na 11 Mg Magnesium Aluminum 12 Al 13 14 Silicon Phosphorus Sulfur 15 16 1s22p 3p Chlorine 17 122 Argon Potassium Calcium Ar 18 19 1s 2 2p 1s 252 l2s 1s2 1s2 ILs22 1s2 2p 1s2s 2p 1 2s 2p 1s 2s Is2s Ca 20 Scandium Sc 21 Titanium 22 Vanadium 23 Chromium Cr 24 Manganese Iron Mn 25 Fe 26 Cobalt Co 27 Nickel Ni 28 Copper Zinc Gallium Cu 29 Zn 30 Ga 31 1s25 2 1s 25 Germanium Ge 32 Arsenic Selenium As 33 Se 34 1525 1s22 Bromine Br 35 s-4p Krypton Kr 36 Metal Key 29 Cu - 63.54 IA Atomic number Nonmetal Symbol H. 1.0080 TIA IIIA IVA VA VIA VIIA 4.0026 Atomic weight 10 Intermediate Li Be Ne 10.811 12.011 14.007 15.999 18.998 20.180 15 6.941 9.0122 11 12 13 14 16 17 18 Na Mg AI Si P. Ar IIIB IVB VB VIB VIIB IB IB 22.990 24.305 26.982 28.086 30.974 32.064 35.453 39.948 23 19 20 21 22 24 25 26 27 28 29 30 31 32 33 34 35 26 Ca 40.08 So Ti 44.956 47.87 Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 50.942 51.996 | 54.938 | 55.845 58.933 72.64 74.922 78.96 79.904 39.098 58.69 63,54 65.4L 69.72 83.80 49 37 38 39 40 41 42 43 44 45 46 47 48 50 51 52 52 54 Rb Sr Nb Mo Te Ru Rh Pd Ag Cd In Sn Sb Te Xe 101.07 102.91 106.4 107.87 112.41| 114.82 | 118.71 | 121.76 | 127.60 126.90 131.30 76 85.47 87.62 88.91 91.22 92.91 95.94 (98) 56 77 79 80 86 55 72 73 74 75 78 81 82 83 84 85 Rare earth Cs Hf Ta Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Ba 178.49| 180.95 183,84 186.2 190.23 132.91 137.24 192.2 195.08 196.97 200.59 204.38 207.19| 208.98 (2091 12100 (222) series Acti- nide 87 88 104 105 106 107 108 109 110 Fr Ra Rf Db Sg Bh Hs Mt Ds (223) (226) (261) 1262) (266) (2641 12771 (268) (2811 ser les 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 Rare earth series La Ce Pr Nd Pm Dy 150.35 | 161.96 167.25 158.92 162.50 | 164.93 167.26 | 168.93 173.04 | 174.97 Sm Eu Gd Tb Ho Er Tm Yb Lu 138.91 | 140.12 140.91 | 144.24 (145 89 90 91 92 94 95 96 97 98 99 100 101 102 103 Lr Actinide series Ac Np 232.04 231.04 | 238.03 Th Pa Pu Am Cm Bk Cf Es Fm Md No (227) 1237) (244) (243) 12471 (247) (2511 12521 (257) (2581 (2591 (262)
Step by Step Solution
3.37 Rating (169 Votes )
There are 3 Steps involved in it
a Electron configuration is that of a transition meta... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
33-E-M-S-E-M-S (11).docx
120 KBs Word File


