A 1.5-kg specimen of a 90 wt% Pb???10 wt% Sn alloy is heated to 250oC (480oF); at
Question:
A 1.5-kg specimen of a 90 wt% Pb???10 wt% Sn alloy is heated to 250oC (480oF); at this temperature it is entirely an a-phase solid solution (Figure). The alloy is to be melted to the extent that 50% of the specimen is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composition while holding the temperature constant.
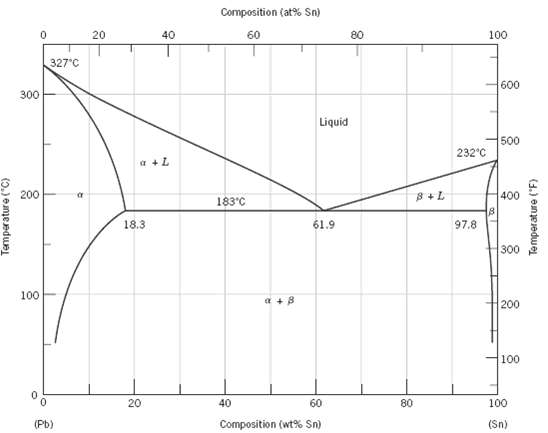
(a)?? ? ??To what temperature must the specimen be heated?
(b)?? ? ??How much tin must be added to the 1.5-kg specimen at 250?C to achieve this state?
Transcribed Image Text:
Composition (at% Sn) 20 40 60 80 100 327°C 600 300 Liquid 500 232°C a +L 200 400 8+L 183'C 18.3 61.9 97.8 300 100 a + B 200 100 20 40 60 80 100 (Pb) Composition (wt% Sn) (Sn) Temperature ("C) (3.) enjesedweL
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a Probably the easiest way to solve this part of the problem is by trial and errorthat is on the PbS...View the full answer

Answered By

Nicholas Maina
Throughout my tutoring journey, I've amassed a wealth of hands-on experience and honed a diverse set of skills that enable me to guide students towards mastering complex subjects. My proficiency as a tutor rests on several key pillars:
1. Subject Mastery:
With a comprehensive understanding of a wide range of subjects spanning mathematics, science, humanities, and more, I can adeptly explain intricate concepts and break them down into digestible chunks. My proficiency extends to offering real-world applications, ensuring students grasp the practical relevance of their studies.
2. Individualized Guidance:
Recognizing that every student learns differently, I tailor my approach to accommodate various learning styles and paces. Through personalized interactions, I identify a student's strengths and areas for improvement, allowing me to craft targeted lessons that foster a deeper understanding of the material.
3. Problem-Solving Facilitation:
I excel in guiding students through problem-solving processes and encouraging critical thinking and analytical skills. By walking learners through step-by-step solutions and addressing their questions in a coherent manner, I empower them to approach challenges with confidence.
4. Effective Communication:
My tutoring proficiency is founded on clear and concise communication. I have the ability to convey complex ideas in an accessible manner, fostering a strong student-tutor rapport that encourages open dialogue and fruitful discussions.
5. Adaptability and Patience:
Tutoring is a dynamic process, and I have cultivated adaptability and patience to cater to evolving learning needs. I remain patient through difficulties, adjusting my teaching methods as necessary to ensure that students overcome obstacles and achieve their goals.
6. Interactive Learning:
Interactive learning lies at the heart of my approach. By engaging students in discussions, brainstorming sessions, and interactive exercises, I foster a stimulating learning environment that encourages active participation and long-term retention.
7. Continuous Improvement:
My dedication to being an effective tutor is a journey of continuous improvement. I regularly seek feedback and stay updated on educational methodologies, integrating new insights to refine my tutoring techniques and provide an even more enriching learning experience.
In essence, my hands-on experience as a tutor equips me with the tools to facilitate comprehensive understanding, critical thinking, and academic success. I am committed to helping students realize their full potential and fostering a passion for lifelong learning.
4.90+
5+ Reviews
16+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
A 90 wt% Ag-10 wt% Cu alloy is heated to a temperature within the + liquid phase region. If the composition of the liquid phase is 85 wt% Ag, determine: (a) The temperature of the alloy (b) The...
-
A Nb-60 wt% W alloy is heated to 2800 C. Determine (a) The composition of the solid and liquid phases in both wt% and at%; (b) The amount of each phase in both wt% and at%; and (c) Assuming that the...
-
A 65 wt% Ni-35 wt% Cu alloy is heated to a temperature within the + liquid-phase region. If the composition of the phase is 70 wt% Ni, determine: (a) The temperature of the alloy (b) The...
-
Anon Amus Inc. operates a woodworking shop that makes tables and chairs. He has 25 employees working 40 hours per week and he has 750 hours per week available in machine time. Ralph knows that he...
-
On December 31, it was estimated that goodwill of $4,000,000 was impaired. In addition, a patent with an estimated useful economic life of 15 years was acquired for $900,000 on August 1. Required: A....
-
1. Which organization structure should Garlick use for ThinkImpact? Explain. 2. If Garlick chooses to create a for-profit entity, either to replace the current nonprofit organization or as a...
-
Describe the different types of events and compare them.
-
Consider Mila Fashion from S8- 5. Assume once again that all fixed costs are unavoidable. If the company discontinues one of the current departments, it plans to replace the discontinued department...
-
Consider this week's Lecture # 1. The three most influential organizations shaping the global economy are World Bank, IMF, and World Trade Organization. - How do their roles differ? - How do wealthy...
-
Howard Poster Incorporated had 12,000 units of work in process in Department A on October 1. These units were 60 percent complete as to conversion costs. Materials are added in the beginning of the...
-
Determine the relative amounts (in terms of mass fractions) of the phases for the alloys and temperatures given in Problem 9.8. (a) 90 wt% Zn-10 wt% Cu at 400(C (750(F) (b) 75 wt% Sn-25 wt% Pb at...
-
A magnesium-lead alloy of mass 5.5 kg consists of a solid phase that has a composition that is just slightly below the solubility limit at 200oC (390oF). (a) What mass of lead is in the alloy? (b)...
-
Can Marta sue Chelene for fraudulent misrepresentation? Why or why not? What element(s) might be lacking? Chelene had been a caregiver for Martas eighty-year-old mother, Janis, for nine years....
-
29. Charles company used the percent of sales method to determine its bad debts expense. At the end of the current year, the company's unadjusted trial balance reported the following selected...
-
The Wagner Corporation has a $23 million bond obligation outstanding, which it is considering refunding. Though the bonds were initially issued at 15 percent, the interest rates on similar issues...
-
Automated Teller Machines (ATM) are designed so that users will provide a personal identification number (PIN) and a card to access their bank accounts. Give examples of confidentiality, integrity...
-
With yields near all time lows, why should investors own bonds? Explain how the article "Why Own Bonds When Yields Are So Low?" (link listed below) helps you better understand some of the more...
-
1) Consider an automated teller machine (ATM) in which users provide a personal identification number (PIN). Discuss what confidentiality, integrity, availability, authenticity, and non-repudiation...
-
Morris Corporation has the following information on its balance sheets: Cash = $40, accounts receivable = $30, inventories = $100, net fixed assets 5 $500, accounts payable = $20, accruals = $10,...
-
A sprinkler head malfunctions at midfield in an NFL football field. The puddle of water forms a circular pattern around the sprinkler head with a radius in yards that grows as a function of time, in...
-
Using heats of formation tabulated in Appendix E, calculate the heats of reaction for the following. (a) C 2 H 2 (g)+ 5/2 O 2 (g) 2 CO 2 (g) + H 2 O() (b) PCl 3 (g) + Cl 2 (g) PCl 5 (g) (c) C 2 H 4...
-
A load of 140,000 N (31,500 lbf) is applied to a cylindrical specimen of a steel alloy (displaying the stress-strain behavior shown in Figure 6.22) that has a cross-sectional diameter of 10 mm (0.40...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
A specimen of copper having a rectangular cross section 15.2 mm 19.1 mm (0.60 in. 0.75 in.) is pulled in tension with 44,500 N (10,000 lbf) force, producing only elastic deformation. Calculate the...
-
It is possible to form an atom out of a muon (like and electron, but with mass approximately given by 105.7 MeV/c 2 ). What would the muonic Bohr radius be? Give a numerical value, and make sure your...
-
Suppose you drop little balls of mass m on to a target marked on the floor. According to the uncertainty principle, the balls should not necessarily fall straight down even if you attempt to hold...
-
First , explain what Social Exchange Theory is and how it functions in Interpersonal Relationships. Second , describe a situation (real or hypothetical) where Social Exchange Theory is a factor in a...

Study smarter with the SolutionInn App


