A 50.0-L tank contains an air?carbon tetrachloride gas mixture at an absolute pressure of 1 atm, a
Question:
A 50.0-L tank contains an air?carbon tetrachloride gas mixture at an absolute pressure of 1 atm, a temperature of 34?C, and a relative saturation of 30%. Activated carbon is added to the tank to remove the CCl4 from the gas by adsorption and the tank is then sealed. The volume of added activated carbon may be assumed negligible in comparison to the tank volume.
(a) Calculate PCCl4 at the moment the tank is sealed, assuming ideal gas behavior and neglecting adsorption that occurs prior to sealing.
(b) Calculate the total pressure in the tank and the partial pressure of carbon tetrachloride at a point when half of the CCl4 initially in the tank has been adsorbed. Note: It was shown in Example 6.7-1 that at 34?C where PCC14 is the partial pressure (mm Hg) of carbon tetrachloride in the gas contacting the carbon.
(c) How much activated carbon must be added to the tank to reduce the mole fraction of CC14 in the gas to 0.001?
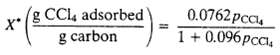
Step by Step Answer:

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau





