(a) Derive the equation relating the molality (m) of a solution to its molarity (M) Where d...
Question:
(a) Derive the equation relating the molality (m) of a solution to its molarity (M)
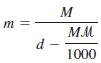
Where d is the density of the solution (g/mL) and m is the molar mass of the solute (g/mol).
(b) Show that, for dilute aqueous solutions, m is approximately equal to M.
Transcribed Image Text:
M m MM d 1000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a If we assume 1 L of solution then we can calculate the mass of solution from its density ...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
85+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
One practical way to measure magnetic field strength uses a small, closely wound coil called a search coil. The coil is initially held with its plane perpendicular to a magnetic field. The coil is...
-
Derive an equation similar to the Henderson-Hasselbalch equation relating the pOH of a buffer to the of its base component.
-
A conveyor drive system to produce translation of the load is shown in Figure. The reducer is a gear pair that reduces the motor speed by a factor of 10:1. The motor inertia is I = 0.002 kg m2. The...
-
List four components and four guidelines that the J. Crew mission statement fails to exhibit. Write a new and improved mission for J. Crew.
-
You purchased your house 5 years ago for $110,000 and based on recent appraisals it can be sold today for $141,000. What effective annual rate of return did you earn?
-
All airplane passengers at the Lake City Regional Airport must pass through a security screening area before proceeding to the boarding area. The airport has three screening stations available, and...
-
Describe an experiment, and give the necessary equations for the measurement of the complex through-thickness shear modulus, \(G_{23}^{*}\), of a unidirectional, specially orthotropic, transversely...
-
A survey of shopping habits found the percentage of respondents that use technology for shopping as shown in Figure 5.37. For example, 17.39% only use online coupons; 21.74% use online coupons and...
-
Determine the cost of a product with a selling price of $45.55, ober head of 5.50$ and net profit of 9$.
-
Big City Gymnastics (BCG or the Club) is a not-for-profit organization that operates a gymnastics club. BCG was incorporated in May 1983 and has operated the Club at the same facility since that...
-
Using Henry's law and the ideal gas equation to prove the statement that the volume of a gas that dissolves in a given amount of solvent is independent of the pressure of the gas.
-
At 298 K, the osmotic pressure of a glucose solution is 10.50 atm. Calculate the freezing point of the solution. The density of the solution is 1.16 g/mL.
-
Construct the empirical cumulative distribution function for the data set of bamboo shoot heights given in DS 6.6.5. Draw 95% confidence bands around the empirical cumulative distribution function....
-
The following data refers to a single cylinder 4 strokes petrol engine. Cylinder diameter \(=30\) \(\mathrm{cm}\), piston stroke \(=40 \mathrm{~cm}\), engine speed \(=1,400\) rpm, indicated mean...
-
A financial institution has rate-sensitive assets of \($392\) million. What is the maximum amount of ratesensitive liabilities this institution will decide to keep in its balance sheet if interest...
-
A four-stroke diesel engine has a piston diameter \(250 \mathrm{~mm}\) and stroke \(400 \mathrm{~mm}\). The mean effective pressure is 4 bar and speed is \(500 \mathrm{rpm}\) the diameter of the...
-
An air at \(15^{\circ} \mathrm{C}\) and 1 bar is compressed adiabatically to 15 bar by an engine working on Otto cycle. The maximum pressure of the cycle is 40 bar. Calculate air standard efficiency,...
-
A gas engine working on 4-stroke cycle has a cylinder diameter \(300 \mathrm{~mm}\) and stroke length of 500 \(\mathrm{mm}\) is running at \(220 \mathrm{rpm}\). Its mechanical efficiency is \(80 \%\)...
-
Is the following statement true or false? The hydrogen atom has a 3s orbital. Explain.
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Ignoring double-bond stereo chemistry, what products would you expect from elimination reactions of the following alkyl halides? Which will be the major product in eachcase? Br CH CH CI CH (a) (b) Br...
-
What alkyl halides might the following alkenes have been madefrom? (b) C CH (a) (a) CH CHH2H2%3DCH2 "CH
-
What stereo chemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1, 2-dibromo-1, 2-diphenylethane? Draw a Newman projection of the reacting conformation?
-
The Power of Trade and Comparative Advantage: Work It Out 3 ? Here's another specialization and exchange problem. This problem is wholly made-up, ? so that you won't be able to use your intuition...
-
Government spending as a fiscal policy tool is used to: A) ?Decrease the national debt B) ?Directly stimulate economic activity by increasing demand C) ?Reduce inflation D) ?Lower interest rates
-
Automobile demand has fallen during a recent recession, and Ana has been laid off from her job on the assembly line. Rajiv recently lost his job as a waiter at a local restaurant. A recent increase...

Study smarter with the SolutionInn App


