(a) Estimate the activation energy for creep (i.e., Qc in Equation 8.20) for the S-590 alloy having...
Question:
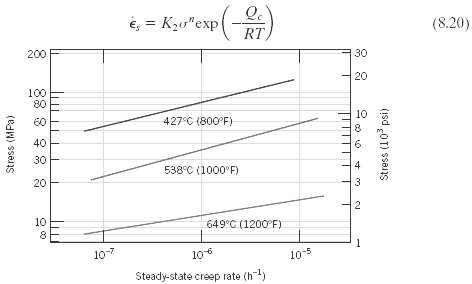
(a) Estimate the activation energy for creep (i.e., Qc in Equation 8.20) for the S-590 alloy having the steady-state creep behavior shown in figure. Use data taken at a stress level of 300MPa (43,500psi) and temperatures of 650°C and 730°C. Assume that the stress exponent n is independent of temperature.
(b) Estimate ?YS at 600°C (873 K) and300MPa.
Transcribed Image Text:
Q. é, = Kzu"exp(-) RT %3D (8.20) 200 30 20 100 80 10 60 427°C (800°F) 40 30 538°C (1000 F) 20 10 649°C (1200 F) 10-7 10 10-5 Steady-state creep rate (h) Stress (MPa) 3. Stress (10° psi)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a We are asked to estimate the activation energy for creep for the S590 alloy having the steadystate ...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
51+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The activation energy for the reaction NO2(g) + CO(g) NO(g) + CO2(g) is 125 kJ/mol, and E for the reaction is 216 kJ/ mol. What is the activation energy for the reverse reaction. [NO(g) + CO2(g) ...
-
The activation energy for the reaction A2(g) + B2(g) 2AB(g) is 167 kJ/mol, and E for the reaction is 128 kJ/mol. What is the activation energy for the decomposition of AB?
-
The activation energy for a certain uncatalyzed biochemical reaction is 50.0 kJ/ mol. In the presence of a catalyst at 37oC, the rate constant for the reaction increases by a factor of 2.50 103 as...
-
Complete the questions in the table below.
-
On November 1 of year 0, Jaxon borrowed $50,000 from Bucksnort Savings and Loan for use in his business. In December, Jaxon paid interest of $4,500 relating to the 12-month period from November of...
-
Consider memory storage of a 32-bit word stored at memory word 42 in a byte-addressable memory.(a) What is the byte address of memory word 42?(b) What are the byte addresses that memory word 42...
-
Explain the three types of visibility for package elements: public, private, and protected.
-
Amortization of Intangibles For each of the following intangible assets , indicate the amount of amortization expense that should be recorded for the year 2010 and the amount of accumulated...
-
You are tasked with predicting the occurrence of a binary event E. That is, = EUE, and ENE 0. Let p and 1-p denote your true probabilities for E and E, respectively. Let r and 1-r denote your...
-
The financial statements of Tarbet Township contain the following data regarding financial position: Governmental fund balances $ 5,000,000 Capital assets used in governmental activities $2,375,000...
-
From Equation 8.19, if the logarithm of is plotted versus the logarithm of ?, then a straight line should result, the slope of which is the stress exponent n. Using Figure 8.31, determine the value...
-
Steady-state creep rate data are given below for nickel at 1000(C (1273 K): If it is known that the activation energy for creep is 272,000 J/mol, compute the steady-state creep rate at a temperature...
-
Some movie studios believe that by adding sexually explicit scenes to the home video version of a movie, they can increase the movies appeal and profitability. A studio executive decided to test this...
-
Suppose a newspaper reporter is diagnosed with epilepsy after having a seizure. She wants to write about the experience and about the specialist doctors and medicines she now needs. She is also...
-
what are the hormonal and cellular mechanisms regulating gametogenesis, folliculogenesis, and spermatogenesis, and how do disruptions in these processes contribute to infertility and reproductive...
-
how do the structural characteristics of the alveolar-capillary membrane facilitate gas exchange, and what factors influence the efficiency of oxygen diffusion and carbon dioxide transport in the...
-
1. Consider the importance of each of these 3 dimensions in predicting what financial service (i.e. debit, credit or saving) could be a better fit with these cultures; could you say what financial...
-
Singer-songwriter Mirella Cesa from Guayaquil, Ecuador, is known as the Mother of Andipop. Watch the music video for Cesa's 2017 single "La corriente" and read more about Mirella Cesa and her music....
-
Explain what is meant by this statement: Our tax rates are progressive.
-
Is it a breach of fiduciary duty for a director of a real estate investment trust (REIT) negotiating a joint venture on behalf of the REIT with another director for the development of a portfolio of...
-
If you are designing a component that needs to be able to change temperature fairly quickly, should you use a material with a high or low specific heat? Explain your answer.
-
Copper (Cu) and platinum (Pt) both have the FCC crystal structure, and Cu forms a substitutional solid solution for concentrations up to approximately 6 wt% Cu at room temperature. Determine the...
-
Calculate the number of vacancies per cubic meter at 1000C for a metal that has an energy for vacancy formation of 1.22 eV/atom, a density of 6.25 g/cm3, and an atomic weight of 37.4 g/mol. (A) 1.49 ...
-
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% Pb and 95.5 wt% Sn? The atomic weights for Pb and Sn are 207.19 g/mol and 118.71 g/mol, respectively. (A) 2.6 at% Pb and...
-
The following balance sheet for the Los Gatos Corporation was prepared by a recently hired accountant. In reviewing the statement you notice several errors. LOS GATOS CORPORATION Balance Sheet At...
-
Using Regression to Calculate Fixed Cost, Calculate the Variable Rate, Construct a Cost Formula, and Determine Budgeted Cost Pizza Vesuvio makes specialty pizzas. Data for the past 8 months were...
-
Mackenzie Corp. is preparing the December 31, 2023, year-end financial statements. Following are selected unadjusted account balances: Estimated warranty liability $ 6,650 Income tax expense Mortgage...

Study smarter with the SolutionInn App


