Question: (a) How would you modify matrices [A]?and [B]?of Example 15.3 if the solution region had charge density ?s? (b) Write a program to solve for
(a) How would you modify matrices [A]?and [B]?of Example 15.3 if the solution region had charge density ?s?
(b) Write a program to solve for the potentials at the grid points shown in Figure assuming a charge density ?s = x(y ??1) nC/m2. Use the iterative finite difference method and take ?r = 1.0.
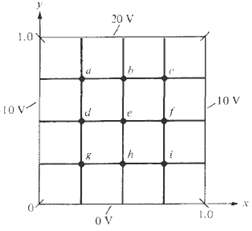
20 V 1.0 10 V 1ov h. 1.0 OV
Step by Step Solution
3.31 Rating (172 Votes )
There are 3 Steps involved in it
a Matrix A remains the same To each term of matrix B we add h 2 s b Th... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
39-P-E-W-A (226).docx
120 KBs Word File


