(a) Is sodium hydroxide a strong enough base to completely remove a proton from the ?-carbon acetone;...
Question:
(a) Is sodium hydroxide a strong enough base to completely remove a proton from the ?-carbon acetone; that is, does this equilibrium lie nearly completely to the right when sodium hydroxide is the base?
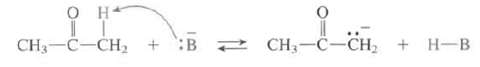
(b) Which common bases can be used to completely remove a proton from acetone?
Transcribed Image Text:
CH3-C-CH2 + :B 2 CH;-C-CH, + H-B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a The equilibrium does not lie completely to the right ...View the full answer

Answered By

Uttam Singh Bhadauriya
I've tutored high school students during my graduation years, and taught them Precalculus and Algebra for college.
I've also taught Calculus and Linear Algebra to College students.
I'm a graduate in Computer Science, but I'm highly enthusiastic about learning and teaching mathematics.
I've qualified exams like IIT-JAM and TIFR, which are entrance exams for research institutes in India.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify which of the following bases can be used to deprotonate a terminal alkyne: (a) NaOCH 3 (b) NaH (c) BuLi (d) NaOH (e) NaNH 2
-
A precipitate forms when a small amount of sodium hydroxide is added to a solution of aluminum sulfate. This precipitate dissolves when more sodium hydroxide is added. Explain what is happening.
-
Sodium hydroxide is hygroscopic-that is, it absorbs moisture when exposed to the atmosphere. A student placed a pellet of NaOH on a watch glass. A few days later, she noticed that the pellet was...
-
9. What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is...
-
Should Wagner be held responsible for these problems? Explain.
-
Thelma and Louise have a big exam tomorrow. Each is trying to decide how much time to spend socializing, and how much to spend studying. We'll use T to stand for the amount of time Thelma spends...
-
0.1 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca 0 z Z .09 .08 .07 .06 .05 .04 .03...
-
Davide's Arrangements purchases, wholesales, and retails fresh flowers. Company estimates reveal the following for the first three months of the company's 2011 fiscal year: Davide's pays 60 percent...
-
1. What is the slope of the Aggregate-Supply curve in the short run? In the long run? What shifts the AS curve(s)? 2. How does the interest-rate effect help explain the slope of the aggregate-demand...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Explain which the most acidic hydrogen's in these compoundsare c) CH,CCH,CH, b) PHCH CCH3
-
Provide names for thesecompounds: b) a) , CI Br - d) ) e) CH3 NO,
-
Why is DNA often described as a double helix?
-
How should the firm be (re)organized internally? In other words, how can internal fit be created between the focal firm's business model and its internal organizational design?
-
Evaluate leadership style which I believe is situational leadership. List three weaknesses, including how each strength relates to situational leadership theory. My weaknesses are delegation, equal...
-
A reaction A B is known to proceed according to the rate expression KICA k2 + CA A pilot-scale CFSTR has been operated at steady state at several different flow rates to determine the
-
One of South Africa's significant trading partners is the European Union (EU). The EU is a major destination for South African exports and a source of imports, contributing significantly to South...
-
Can the exploration of psychological states such as altered consciousness, dreams, and subconscious realms unlock new avenues of artistic inspiration, transcending conventional boundaries of...
-
Predicting Prices of Used Cars (Regression Trees). The file ToyotaCorolla.csv contains the data on used cars (Toyota Corolla) on sale during late summer of 2004 in the Netherlands. It has 1436...
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
Of the nearly 1,300 species officially classified as endangered under the 1973 Endangered Species Act, between 450 and 500 are usually considered to be stable or improving. The remaining species are...
-
The following model is that of cholic acid, a constituent of human bile. Locate the three hydroxyl groups, and identify each as axial or equatorial, is cholic acid an A t trans steroid or an A-B...
-
Propose a biosynthetic pathway for the sesquiterpene helminthogermacrene from farnesvldiphosphate.
-
Identify the following fatty acid, and tell whether it is more likely to be found in peanut oil or in redmeat:
-
State the dividend irrelevance proposition. What are the assumptions behind this proposition? Explain why this proposition does not hold in the real world. (20 marks)
-
How many monthly withdrawals of $1,400 will an investment of $75,000 sustain if the first withdrawal is made 12 months from now and the money earns 8.4% compounded monthly
-
1. You buy a bond with 3 years left to maturity and a yield to maturity of 6% for $920. After 1 year you receive a coupon payment of $30 and sell the bond for $940. What was your rate of return on...

Study smarter with the SolutionInn App


