Question: Explain which the most acidic hydrogen's in these compoundsare c) CH,CCH,CH, b) PHCH CCH3
Explain which the most acidic hydrogen's in these compoundsare
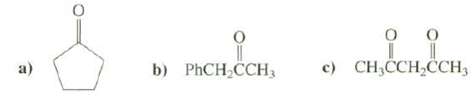
c) CH,CCH,CH, b) PHCH CCH3
Step by Step Solution
3.49 Rating (162 Votes )
There are 3 Steps involved in it
a The circled hydrogens are more acidic because the conjugate base is stab... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-N (55).docx
120 KBs Word File


