Question: A toroid with N turns carrying a current I has mean radius R and cross-sectional radius r, where r R
A toroid with N turns carrying a current I has mean radius R and cross-sectional radius r, where r
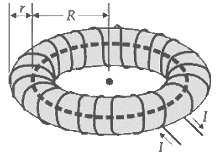
R
Step by Step Solution
3.41 Rating (164 Votes )
There are 3 Steps involved in it
For a toroid B app 0 NI ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
10-P-E-M-F (885).docx
120 KBs Word File


