Addition of HBr to a double bond with an ether (?OR) substituent occurs region specifically to give
Question:
Addition of HBr to a double bond with an ether (?OR) substituent occurs region specifically to give a product in which the ?Br and ?OR are bonded to the same carbon. Draw the two possible carbocation intermediates in this electrophilic addition reaction, and explain using resonance why the observed product is formed.
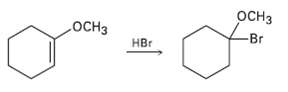
Transcribed Image Text:
осHз оСнз HBr -Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
OCH 3 0 Br H OCH 3 HBr OCH3 H or H OCH3 ...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced. Br + HBr
-
In the addition of HBr to 3,3-dimethyl-l-butene, the results observed are shown in Fig. P5.36. (a) Explain why the different conditions give different product distributions. (b) Write a detailed...
-
Predict the major product from addition of HBr to each of the following alkenes: (a) CH2 (b) (c) CH CH3CHHHH
-
Locate the centroid of the region bounded by the given curves. about x = 5, y = 2x2, y = 0, x = 5 < UseHorizontal ElementOf Area > x = 5 y = 2x? y = 0
-
Refer to the Big Rig Rental Company case. Design a spreadsheet that will allow the firm to determine the Net Present Value of cash flows over the five-year period. The following exercises refer to...
-
Apply source transformation to determine vo and io in the circuit in Fig. 4.89. 12 V F 2A
-
Jo, John and Jack are three generations of the one family involved for nearly 50 years in providing legal services. The firm is preparing its fees budget for the year ending 30 June 2025. It budgets...
-
Garmen Technologies Inc. operates a small chain of specialty retail stores throughout the U.S. Southwest. The company markets technology-based consumer products both in its stores and over the...
-
Shadee Corporation expects to sell 600 sun shades in May and 800 in June. Each shade sells for $180. Shadee's beginning and ending finished goods inventories for May are 75 and 50 shades,...
-
You have estimated the following cash flows on a project: Year Cash Flow to Equity ($) 0 .. 5,000,000 1 .. 4,000,000 2 .. 4,000,000 3 .. 3,000,000 Plot the NPV profile for this project. What is the...
-
Why do you suppose it?s not possible to prepare a Grignard reagent from a bromo alcohol such as 4-bromo-1-pentanol? Give another example of a molecule that is unlikely to form a Grignard reagent....
-
Alkyl halides can be reduced to alkanes by a radical reaction with tributyltin hydride, (C 4 H 9 ) 3 SnH, in the presence of light (h v ). Propose a radical chain mechanism by which the reaction...
-
For each equation in Exercises, under the given condition: (a) Find k and (b) find a second solution. 1. kx2 - 17x + 33 = 0; one solution is 3 2. kx2 - 2x + k = 0; one solution is -3
-
Year Revenues 1 $ 50,000 2 3 4 Thereafter 40,000 20,000 10,000 Expenses are expected to be 50% of revenues, and working capital required in each year is expected to be 20% of revenues in the...
-
What are the two ways for primary governments to report component units?
-
Schrute Farms has $ 5 3 , 0 0 0 in fixed assets at the beginning of the year during the year the company purchased $ 1 1 , 4 0 0 in new equipment. It also sold out a price of $ 2 0 0 0 from old...
-
Name at least 2 types of leadership styles and provide a brief explanation of each. 2. Research and contrast the leadership styles of the late Apple Inc. Cofounder Steve Jobs and the current CEO...
-
Riverbed Services Inc. issued 1,000 $2 convertible preferred shares at $75 and 5,600 common shares at $25 each in 2022. Each preferred share is convertible into three common shares. On March 15,...
-
Using workplace examples, contrast diversity, equity, and inclusion.
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
Draw the Lewis structure for each compound. a. Cl 2 O 7 (no ClCl bond) b. H 3 PO 3 (two OH bonds) c. H 3 AsO 4
-
When optically pure (R)-2-bromobutane is heated with water, butan-2-ol is the product. The reaction forms twice as much (S)-butan-2-ol as (R)-butan-2-ol. Calculate the e.e. and the specific rotation...
-
A chemist finds that the addition of (+)-epinephrine to the catalytic reduction of butan-2-one (Figure 5-16) gives a product that is slightly optically active, with a specific rotation of +0.45o...
-
1. Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity. (a) 1-bromo-1-chloroethane (b) 1-bromo-2-chloroethane...
-
How is decision-making in organizations best supported by Management Accounting (a descriptive nature) and how should it be supported by Management Accounting (a prescriptive nature)? Is there a gap...
-
Discuss the thermodynamic principles governing the separation of azeotropic mixtures using advanced distillation techniques such as pressure swing distillation, extractive distillation, and hybrid...
-
respond to the discussion In the IMA article, Activity-Based Costing (ABC) is explained as a cost allocation technique that traces indirect costs (often referred to as "overhead") to specific cost...

Study smarter with the SolutionInn App


