Question: Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction,
Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:
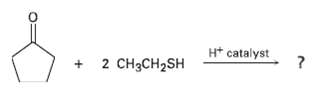
H* catalyst 2 CH3CH2SH
Step by Step Solution
3.35 Rating (167 Votes )
There are 3 Steps involved in it
The same series of steps used to form an acetal is followed in thi... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-AN-96.docx
120 KBs Word File


