Alkyl benzenes such as toluene (methylbenzene) react with NBS to give products in which bromine substitution has
Question:
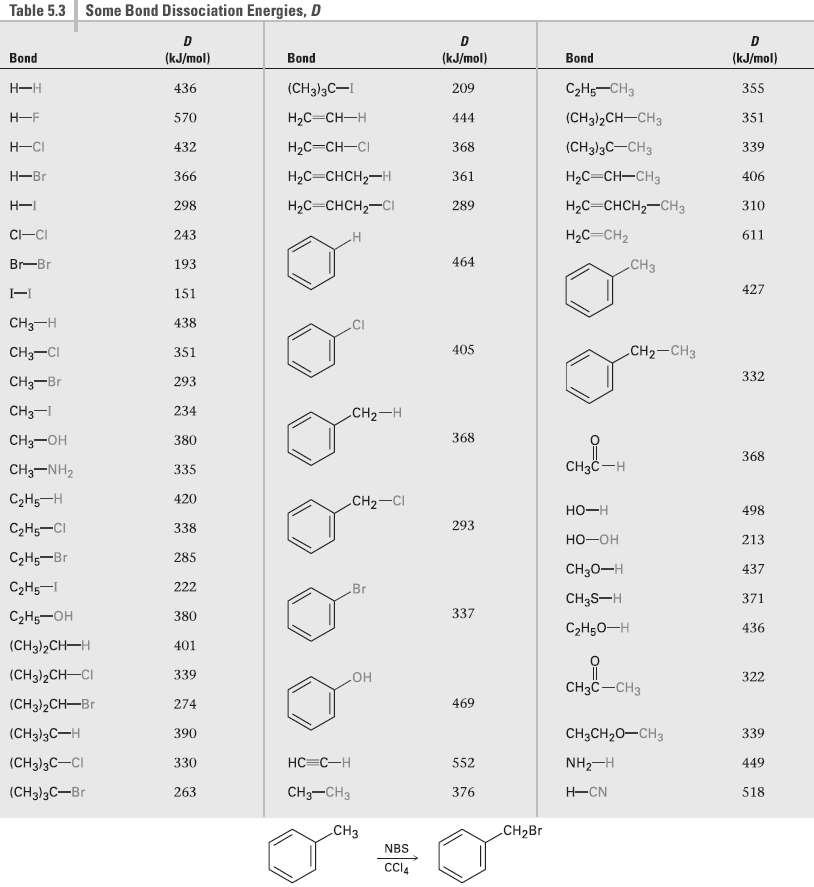
Alkyl benzenes such as toluene (methylbenzene) react with NBS to give products in which bromine substitution has occurred at the position next to the aromatic ring (the benzylic position). Explain, based on the bond dissociation energies in Table 5.3.
Transcribed Image Text:
Table 5.3 Some Bond Dissociation Energies, D (kJ/mol) (kJ/mol) (kJ/mol) Bond Bond Bond С-На —снз (CH3)3C-I Н—н 436 209 355 (CHg]2CH -CHз Н-F НаС —Сн—н 570 444 351 НаС— Сн—CI Н-СI (CH3)3C-CH3 432 368 339 Hас — снсH2—н Hас — Сн—сна Н—Br 366 361 406 Нас снсн—снз Hас - снсH2—сI Н- 298 289 310 Hас Сн2 C-CI 243 611 464 CHз Br-Br 193 427 151 CH3-H 438 CI CH— CHз 405 CH3-CI 351 332 CHз—Br 293 CHз-1 234 CH2-H CHз—он 368 380 368 Cнзс —н CHз-NH2 335 СаНs —Н 420 CH2-CI 498 но-н 293 СНо—СI 338 но-он 213 СаН — Br 285 CH30-H 437 СаНs — 222 Вг CH3S-H 371 337 СэН—он 380 СэHоо—н 436 (CH3)2Cн—н 401 (CH3)2CH-CI 339 322 CнзC —CHз но 469 (CH3)2CH-Br 274 (CH])3C—н 390 CH;CH20-CH3 339 НC -С —Н (CHд)3C—CI 330 NH2-H 552 449 CH—CHз H-CN (CH3)3C-Br 263 376 518 .CHз CH2B NBS CCI4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (12 reviews)
Table 53 shows that the bond dissociation energy of C 6 H 4 CH 2 H is 368 kJmol This value is compar...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Basing your answers on the bond dissociation energies in Table 4.3, calculate which of the following reactions are endothermic and which are exothermic: (a) (CH3)2CHOH + HF - (CH3)2CHF + H2O (b)...
-
Find examples of C-H bond dissociation energies in Table 10.1 that are as closely related as possible to the bonds to Ha, Hb, and Hc in the molecule at right. Use these values to answer the questions...
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
Write MATLAB code of the question. Do all parts and show code with comments and also attach a screenshot of code and outputs. Using the results of Problem 4.13, verify the following properties of WN...
-
a. Consider a recommendation that the prices for the Educational, Large-Scale, and High-Speed versions should be $75, $275, and $475, respectively. What annual profit would these prices achieve? b....
-
Find the nodal voltage v1 through v4 in the circuit in Fig. 3.120 using PSpice. Figure 3.120 6/ 22 | 12 8 A 4 S2 212 20 v
-
Eastern Engines Ltd has a policy of maintaining a finished goods inventory balance at the end of a month equal to 25% of the sales requirements of the following month. The ending inventory balance on...
-
Lei Materials balance sheet lists total assets of $1 billion, $100 million in current liabilities, $400 million in long-term debt, $500 million in common equity, and 50 million shares of common...
-
Discuss the role of real-time communication protocols in RTOS-based systems. How do these protocols ensure real-time data exchange while maintaining system stability and predictability ?
-
Calm Day reported the following income statement for the year ended December 31, 2019: Requirements 1. Compute Calm Day's inventory turnover rate for the year. (Round to two decimal places.) 2....
-
What product(s) would you expect from the reaction of 1, 4-hexadiene with NBS? What is the structure of the most stable radical intermediate?
-
Draw resonance structures for the benzyl radical, C6H5CH12, the intermediate produced in the NBS bromination reaction of toluene.
-
For each of the following items, circle either a or b. In some cases, both a and b may apply to you. You should decide which is more like you, even if it is only slightly more true. I like to a. Work...
-
What are psychologist highlighted the issues with psychological diagnosis?
-
Question 1 [10 points] Write a Makefile that builds a C program map.c and produces the executable object file map. Your Makefile should use the compiler flags discussed in class, and have the 'all'...
-
What strategic value do middle managers have for companies? 2. What are reasons that companies doubt the value of middle managers? 3. Summarize in your own words all the steps that google took to...
-
Date CAT MOT 01-Dec-05 52.09 21.51 01-Nov-05 52.1 22.9 03-Oct-05 47.42 21.06 01-Sep-05 52.74 20.94 01-Aug-05 49.81 20.76 01-Jul-05 48.39 20.1 01-Jun-05 42.58 17.33 02-May-05 42.04 16.45 01-Apr-05...
-
How can define Great man theory, Transformational Theory and Visionary Leadership ?
-
Does an organization have an ethical responsibility to provide career development advice to employees? Explain your position.
-
Chicago Company sold merchandise to a customer for $1,500 cash in a state with a 6% sales tax rate. The total amount of cash collected from the customer was $558. $600. $642. $636. Nevada Company...
-
Consider the generic ionic compounds with the formulas AX and BY and the following solubility rules: AX soluble; BY soluble; AY soluble; BX insoluble Let circles represent A+ ions; squares represent...
-
In the presence of a small amount of bromine, the following light-promoted reaction has been observed. (a) Write a mechanism for this reaction. Your mechanism should explain how both products are...
-
For each compound, predict the major product of free-radical bromination. Remember that bromination is highly selective, and only the most stable radical will be formed. (a) Cyclo-hexane (b)...
-
When exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a chlorination reaction occurs. The products are found to contain substantial amounts of...
-
A project requires an initial investment of $100,000 to purchase equipment. The equipment will be depreciated on a straight-line basis over the five-year life of the project. The company expects the...
-
Vodaphone's earnings per share in 2022 was $1.80, and in 2017 it was $1.25. The company distributes 60% of its earnings as dividends, and the stock is currently valued at $37.75. The cost to issue...
-
A bond is currently selling in the market for $1,085.96. It has a coupon of 8% and a 15-year maturity. Using annual compounding, what is the yield to maturity on this bond?

Study smarter with the SolutionInn App


