An electrospray/transmission quadrupole mass spectrum of the -chain of hemoglobin from acidic solution exhibits nine peaks corresponding
Question:
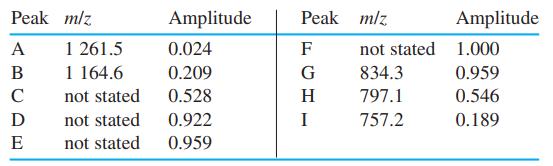
An electrospray/transmission quadrupole mass spectrum of the α-chain of hemoglobin from acidic solution exhibits nine peaks corresponding to Mn+n. Find the charge, n, for peaks A-I. Calculate the molecular mass of the neutral protein, M, from peaks A, B, G, H, and I, and find the mean value.
Transcribed Image Text:
Peak m/z Amplitude Peak mlz Amplitude 1 261.5 1 164.6 A 0.024 F not stated 1.000 В 0.209 G 834.3 0.959 C not stated 0.528 H 797.1 0.546 D not stated 0.922 I 757.2 0.189 E not stated 0.959
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The analysis follows the same steps as Table 213 The work is set out in t...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a...
-
Cocaine metabolism in rats can be studied by injecting the drug and periodically withdrawing blood to measure levels of metabolites by HPLC-mass spectrometry. For quantitative analysis, isotopically...
-
Carbon has two stable isotopes, 126C and 136C, and fluorine has only one stable isotope, 199F. How many peaks would you observe in the mass spectrum of the positive ion of CF+4? Assume that the ion...
-
What approaches to recruitment can an employer adopt in order to create and project a positive public image?
-
Buffet Appliance Mart began operations on May 1. It uses a perpetual inventory system. During May, the company had the following purchases and sales for its Model 25 Sureshot camera. Instructions (a)...
-
Help for Insomniacs A recent study shows that just one session of cognitive behavioral therapy can help people with insomnia. In the study, forty people who had been diagnosed with insomnia were...
-
Vibration spectra can have many frequency peaks. What is key to simplifying the analysis of the number of peaks of interest?
-
The Mainor School District is about to establish a 30-machine computer lab. It is considering six alternative means of acquiring and nancing the machines: 1. Buy the machines outright; cost will be...
-
(1 point) A cryptography student has accidentally used the same keystream to encrypt two different messages, M and M2. We know that the ciphertexts are C 0x06c6d00e4633400a and C2 =...
-
GV is a small accounting firm supporting wealthy individuals in their preparation of annual income tax statements. Every December, GV sends out a short survey to its customers, asking for the...
-
(a) To detect the drug ibuprofen by liquid chromatography/ mass spectrometry, would you choose the positive or negative ion mode for the spectrometer? Would you choose acidic or neutral...
-
Define the unit dalton. From this definition, compute the mass of 1 Da in grams. The mean of 60 measurements of the mass of individual E. coli cells vaporized by MALDI and measured with a quadrupole...
-
Write an equation for each parabola. 2 (2, 1) -2 (1, 0) -2
-
For transaction three, does the amount in the payroll register report match the balance in the payroll account before the bank transfer?
-
Why might venture capitalists choose to invest in a start-up's convertible preferred stock instead of in: a) debt; b) non-convertible preferred stock; or c) common stock (equity)? Explain your...
-
The window shown here would be used in QuickBooks Online to perform maintenance on what type of record?
-
What transaction-related objective is described by ensuring that recorded purchase transactions represent inventories acquired during the period?
-
You have replaced the former audit partner Brett of the auditing firm Moira & Stella (M&S). You find the following independent engagement is potentially in breach of the independence requirements of...
-
Explain why it does not make sense to find a least-squares regression line for the Bear Market data from Problem 34 in Section 4.1. Data from in problem 34 A bear market in the stock market is...
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
Air pollution in the Mexico City metropolitan area is among the worst in the world. The concentration of ozone in Mexico City has been measured at 441 ppb (0.441 ppm).Mexico City sits at an altitude...
-
From the data in Table 18.1, calculate the partial pressures of carbon dioxide and argon when the total atmospheric pressure is 1.05 bar.
-
The average concentration of carbon monoxide in air in an Ohio city in 2006 was 3.5 ppm. Calculate the number of CO molecules in 1.0 L of this air at a pressure of 759 torr and a temperature of 22oC.
-
Individual Retirement Account (IRA) Bonds Mutual fund Stocks Futures Defined contribution plans What is it? Level of Risk and Potential Return Minimum investment? Easy to start or stop?
-
1. A company purchased machinery in 2015 for $400,000. Its value in 2018 was $320,000. Assuming the resale value decreases exponentially, what will the value be in 2020? As a part of your solution,...
-
ROA of a company is 8.57%, Total assets end of the year of 2021 are $9.6 million, ROE is 14% and Profit margin of 19.9% what is the firms value of net income? and what is stockholders equity?

Study smarter with the SolutionInn App


