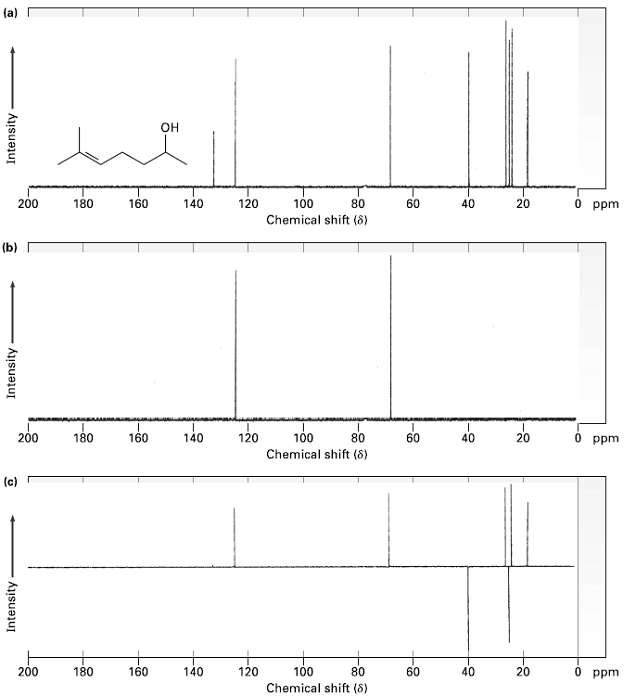
Assign a chemical shift to each carbon in 6.methyl-5-hepten-2-ol(figure). (a) OH 0 ppm 200 180 160 140
Question:
Assign a chemical shift to each carbon in 6.methyl-5-hepten-2-ol(figure).
Transcribed Image Text:
(a) OH 0 ppm 200 180 160 140 120 100 80 60 40 20 Chemical shift (8) (b) 200 180 120 160 100 60 140 80 0 ppm Chemical shift (8) (c) 160 200 180 140 120 100 Chemical shift (8) 80 60 40 20 0 ppm Intensity Intensity Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
Strategy The top spectrum shows all eight 3C NMR peaks The middle spectrum DEPT90 shows only pea...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance: (b) NHCH3 Adrenaline Estrone (a hormone)
-
Carbon tetrachloride is an ozone-depleting chemical found in the atmosphere. The table below shows the concentration of the chemical in parts per trillion (ppt) measured in the European Union. a....
-
Assign oxidation states to each carbon of ethanol, acetaldehyde, and acetic acid. [Ol Ethanol Acetaldehyde Acetic acid
-
Runnals National Bank has experienced the following trends over the past five years (all figures in millions of dollars): Input Area: 1 2 3 4 5 Net Income (after tax) 2.65 2.75 3.25 3.65 4.00 Total...
-
Researchers have found that industries with high entry rates tended to also have high exit rates. Can you explain this finding? What does this imply for pricing strategies of incumbent firms?
-
If the correlation is positive, the slope of the regression line is ____.
-
If a nurse disagrees with a physicians written orders, discuss what action the nurse should take to protect the patients safety.
-
Loco Leasing and Manufacturing Company uses leases as a means of financing sales of its equipment. Loco leased a machine to Potomac Construction for $15,000 per year, payable in advance, for a...
-
Calculate the weighted average cost of debt from the following information. Debentures paying a coupon interest rate of 9%, with a current market value of $1.5 million, have a current market yield of...
-
The guidance system of a ship is controlled by a computer that has three major modules. In order for the computer to function properly, all three modules must function. Two of the modules have...
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
Estimate the chemical shift of each carbon in the following molecule. Predict which carbons will appear in the DWPT-90 spectrum, which will give positive peaks in the DEPT-135 spectrum, and which...
-
The chief financial officer for Eagles Beach Wear and Gift Shop is planning for the companys cash flows for the next six months. The following table summarizes the expected accounts receivables and...
-
How do metabolic flux analysis techniques, such as isotopic labeling, metabolomics, and flux balance analysis, provide quantitative insights into metabolic pathway activity, metabolic flux...
-
What are the metabolic adaptations underlying cancer metabolism, including the Warburg effect, glutamine addiction, and alterations in nucleotide metabolism, and how do these metabolic rewiring...
-
Cash Flow for ABC Hotel July August September Opening Balance $ 85,000 $ 88,450 ? Cash Incoming Sales $ 67,000 $ 64,399 $ 62,500 Asset Sales $ 1,200 $ - $ - Debtor Receipts $ 2,500 $ 2,800 $ 1,300...
-
Can you explain what affiliate marketing fees is? And can you give examples of companies who use it?
-
what ways does the endocrine system interact with other physiological systems, including the nervous system, immune system, and reproductive system, to coordinate complex physiological responses,...
-
A study was conducted where 36 patients were randomly assigned to use one of the three over-the-counter pain medications and were asked how long the medicine provided pain relief (rounded to the...
-
Which of the following raises the credibility of areport? Which of the following raises the credibility of a report? Multiple Choice avoiding predictions avoiding the use of cause-effect statements...
-
Determine the required concentration (in percent by mass) for an aqueous ethylene glycol (C 2 H 6 O 2 ) solution to have a boiling point of 104.0 C.
-
When 1-cyclohexylethanol is treated with concentrated aqueous HBr, the major product is 1-bromo-1-ethylcyclohexane. (a) Propose a mechanism for this reaction. (b) How would you convert...
-
Show how you would make each compound, beginning with an alcohol of your choice. (a) (b) (c) (d) (e) (f) (g) (h) CHO CH,Br CI CH CH 1 CH C OH CH3 OTs
-
Predict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents. (a) PBr3 (b) SOCI2 (c) Lucas reagent (d) Concentrated HBr (e) TsCI/pyridine,...
-
1. Mention five key organizational characteristics that the Cll research team identified for organizations with effective quality management systems. provide brief discussion of each and how it...
-
When a metal was exposed to photons at a frequency of 1.46 1015 s1, electrons were emitted with a maximum kinetic energy 3.60 10-19 J. G ? Calculate the work function, , of this metal. J/photon What...
-
No. 101 Debit Cash $ 2,400 Question 2 (10 pts) On November 1, 2024, the account balances of Schilling Equipment Repair were as follows. Accumulated Depreciation-Equipment A Clipboard Font Alignment...

Study smarter with the SolutionInn App


