Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and
Question:
Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and biologically potent of mammalianprostaglandin
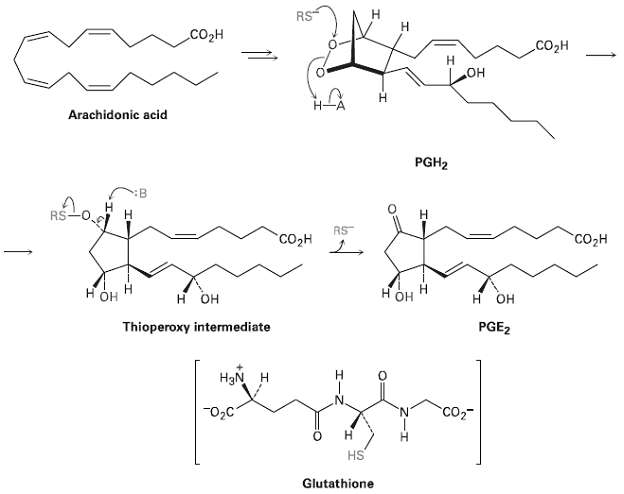
Transcribed Image Text:
н Н RS Соон .CO2H он Н Arachidonic acid PGH2 H. RS RS "СОдн "Соон нон он H OH OH PGE2 Thioperoxy intermediate HgN H "02C CO2 н HS Glutathione
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
R H I R ...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to each chirality center in the followingmolecules: (b) NH2 (a) . "C r "O
-
Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each is a D sugar or an L sugar: (b) (a) - (c) - - - - - - - - - - CH- CH2 CH2
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
The management accountant is preparing the master budget for her retail firm. The following information has been supplied Sales $300,000 Opening inventory $40,000 Closing inventory $60,000 Required...
-
How does an agenda help make a meeting more successful?
-
You charge up the Van de Graaff generator shown in Fig. 22.26, and then bring an identical but uncharged hollow conducting sphere near it, without letting the two spheres touch. Sketch the...
-
Consider the simple linear regression model fit to the weight and blood pressure data in Problem 2.10. Problem 2.10 The weight and systolic blood pressure of 26 randomly selected males in the age...
-
OS Environmental provides cost-effective solutions for managing regulatory requirements and environmental needs specific to the airline industry. Assume that on July 1, 2015, the company issues a...
-
Accounting for Fair Value Hedge: Interest Rate Swap On January 1 of Year 1, Innovative Lab issued a 4-year $50,000 note to a local bank with fixed interest payments based on 6%, payable annually on...
-
The paralegal is assigned the task of preparing an office legal memorandum. The memorandum is due in 10 days, and there is a 5-page limit. The facts and law are as follows: Facts: Mary was Tom's...
-
Write the saponification reaction of glyceryl di-oleate monopalmitate with aqueous NaOH.
-
Studies of the conversion of mevalonate 5-phosphate to isopentenyl diphosphate have shown the following result. Which hydrogen, pro-R or pro-S, ends up cis to the methyl group, and which ends...
-
How do groups handle risk? What is a risky shift? What is a conservative shift?
-
Question 1 Shauna Sullivan's son Simon, age nine, has been bullied at school because he has dyslexia. It all started when a supply teacher asked him to read a passage aloud in class. Since that time,...
-
1. Describe the evidence rule with the most significant impact on the outcome of criminal matters. 2. Provide examples of federal statutes in contract law. 3. Explain and describe the concept of...
-
Ria works at a Quickie Carwash. That morning she was reluctant to go into work because her young son had complained of feeling hot and dizzy and she was concerned that he might be getting sick, but...
-
DIY is a large home improvement chain with stores in the southeastern United States. It sells lumber, hardware, paint and other products typical of such a business. A majority of its sales are to...
-
RES501 Instructions Read the situation, then complete the two parts and associated tasks that follow. Submit all your answers in one document. Write the number of each question you are answering....
-
For the system of Problem 7.2-5 and Fig. P7.2-5: (a) Plot the \(z\)-plane root locus. (b) Plot the \(w\)-plane root locus. (c) Determine the range of \(K\) for stability using the results of part...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
A 0.148 M solution of a monoprotic acid has a percent ionization of 1.55%. Determine the acid ionization constant (K a ) for the acid.
-
Describe the 1H NMR spectrum of a. BrCH2CH2Cl b. ClCH2CH2Cl
-
Write an equation for the reaction of pyridine with a. cold sulfuric acid (H2SO4) b. cold nitric acid (HNO3)
-
Although nitration of pyridine requires a temperature of 300°C (eq. 13.2), 2,6 dimethylpyridine is readily nitrated at 100°C. Write an equation for the reaction, and explain why milder...
-
A portfolio has a standard deviation of 25%. The correlation of the portfolio and the market is 1. If the risk-free rate is 3.2%, the expected return on the market portfolio is 11%, and the standard...
-
(6 pts.) The following gas phase reaction system is conducted in an isothermal constant volume batch reactor. The reactor is charged with pure A with an initial concentration CAO AB ri = k CA B D r2...
-
Based on the NPV and IRR investment criteria, should your company introduce a new candy flavor? 1) Should the cost of a marketing survey be included in your decision? Why or why not? 2) Calculate the...

Study smarter with the SolutionInn App


