Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each
Question:
Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each is a D sugar or an L sugar:
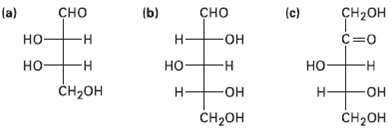
Transcribed Image Text:
(b) (a) но сно сно ҫн-он (c) -н н -Он но- -н но- -н но- -н н- н н но- CH-он CH2он Он CH2он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
The hydroxyl group bonded to the chiral carbon farth...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to each chirality center in the followingmolecules: (b) NH2 (a) . "C r "O
-
Assign R or S configuration to each chirality center in prostaglandin E2 (Figure) the most abundant and biologically potent of mammalianprostaglandin RS .CO2H Arachidonic acid PGH2 H. RS RS " " ...
-
Assign the configuration of each chirality center in the following compounds: (a) (b) (c) (d) (e) C=0 - - - H,OH %3D - - - H,OH
-
Consider the following scenario. Each day, a butcher buys a 200-kilogram pig for $360. The pig can be processed to yield the following three products: Day 1 The butcher buys a pig. The $360 joint...
-
What steps can you take to ensure success with online presentations?
-
For each of the following pairs of semiconductors, decide which will have the smaller band gap energy, Eg, and then cite the reason for your choice. (a) ZnS and CdSe. (b) Si and C (diamond). (c)...
-
A chemical engineer found that by adding different amounts of an additive to gasoline, she could reduce the amount of nitrous oxides (NOx) coming from an automobile engine. A specified amount will be...
-
Grass Security, which began operations in 2015, invests in long-term available-for-sale securities. Following is a series of transactions and events determining its long-term investment activity....
-
Critically discuss, how political and cross-cultural factors can influence IHRM practices in MNC'S. Use examples to substantiate your discussion.
-
Draw a diagram similar to Figure 1.1 that places the following words in the proper relationships: theory, hypothesis, model, data, guess, and law. Figure 1.1 Geology Geochemistry (chemistry of the...
-
Redraw the following aldotetrose as a Fischer projection, and assign R or S configuration to each chiralitycenter:
-
(+)-Arabinose, an aldopentose that is widely distributed in plants, is systematically named (2k, 35, 45)-2, 3, 4, 5-tetrahydroxypentanal. Draw a Fischer projection of (+)-Arabinose, and identify it...
-
An examination designed to measure basic knowledge of American history was given to random samples of freshmen at two major universities, and their grades were Use the U test at the 0.05 level of...
-
Analyzing Alcohol and Tobacco Advertising . Select a recent advertisement for alcohol and/or tobacco from a magazine or online source, paste it into your assignment and answer the following...
-
What is Managerial leadership perspective what are the essential elements that included in Managerial leadership what are the advantages and disadvantages of Managerial leadership
-
Find an organizational chart for your employer or a company that you are familiar with. Is it primarily functional or process-focused? Explain.
-
Name Due Date Assignment Solutions to the quadratic equation Ax +Bx + C = 0 may be calculated using the quadratic formulas: x = -B+B2-4AC 2A -B-B-4AC 2A These formulas may be used, of course, only if...
-
Explain positional bargaining strategy between management and union on multi-shift operations in manufacturing companies
-
You want to withdraw a single sum amount of $6,000 from an account at the end of 7 years. This withdrawal will zero out the account. What single sum of money deposited today is required if the...
-
A company has the following incomplete production budget data for the first quarter: In the previous December, ending inventory was 200 units, which was the minimum required, at 10% of projected...
-
Define the acid ionization constant and explain its significance.
-
In the "A Word About . . . Polycyclic Aromatic Hydrocarbons and Cancer" on page 136, it was noted that some polycyclic aromatic hydrocarbons are used to treat cancer. Indeed, daunorubicin (below) is...
-
Bromination of anthracene gives mainly 9-bromoanthracene. Write out the steps in the mechanism of this reaction. Br 9-bromoanthracene
-
Draw the structures for ortho-xylene and meta-xylene.
-
es Hart, Attorney at Law, experienced the following transactions in Year 1, the first year of operations: 1. Accepted $16,600 on April 1, Year 1, as a retainer for services to be performed evenly...
-
Dahlia Corporation has a current accounts receivable balance of $439,516. Credit sales for the year just ended were $5,503,810. a. What is the receivables turnover? Note: Do not round intermediate...
-
Why does the organizational structure hold political significance? Provide an in-depth analysis of this concept using examples from both academic literature and real-world instances. Additionally,...

Study smarter with the SolutionInn App


