Question: Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in figure. At what positions in mevalonate would the isotopic labelappear?
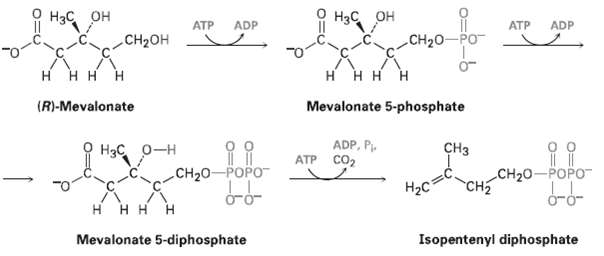
, H3C OH ATP ADP ATP ADP CH20-PO CH2OH H H H H Mevalonate 5-phosphate (R)-Mevalonate ADP, P. ATP Co2 CH H - CH20-POPO CH2 CH20-POPO- H2C H H H H Mevalonate 5-diphosphate Isopentenyl diphosphate
Step by Step Solution
3.36 Rating (159 Votes )
There are 3 Steps involved in it
COAS CH3 COAS CH3 H3COH ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-B-M (158).docx
120 KBs Word File


