Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as
Question:
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in squalene where the label wouldappear.
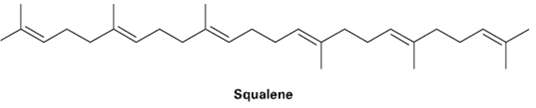
Transcribed Image Text:
Squalene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Farnesyl diphosphate ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that acetyl COA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in lanosterol where...
-
Identify the carbon atoms expected to be labeled with 14C when each of the following substances is biosynthesized from acetate enriched with 14C in its methyl group: (a) (b) (c) (d) Palmitic acid CH3...
-
Label each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°). (a) CH3 CH2 CH (CH3)2 (b) (CH3)3 CCH2CH3 (c) (CH3)2 CHCH (CH3) CH2 CH3 (d) (e) (f) ...
-
7. Please allocate the following terms to the different dimensions Heart sounds, volume, magnetic field changes, pressure, length, extinction/absorption, area, temperature, potential, partial...
-
As a strategic leader, what actions could you take to establish and emphasize ethical practices in your firm? Discuss.
-
A risk profile is constructed in a way that is conceptually similar to performing a _____. Multiple choice question. net present value (NPV) analysis scenario analysis sensitivity analysis Monte...
-
Show that $\operatorname{Var}(\hat{\mathbf{y}})=\sigma^{2} \mathbf{H}$.
-
The original STRIPS program was designed to control Shakey the robot Figure shows a version of Shakeys world consisting of four rooms lined up along a corridor, where each room has a door and a light...
-
Blue Industries purchased a machine from Calico Corporation on October 1, 2016. In payment for the $144,000 purchase, Blue issued a one-year installment note to be paid in equal monthly payments at...
-
Clear Lake Nursing Home defines outputs as a patient day. Its present volume is 26,000 patient days. The average cost per day is $90,00. Present revenues and costs are presented below: Clear Lake...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in ?-cadinol where...
-
Propose a mechanistic pathway for the biosynthesis of caryophyllene, a substance found in cloveoil. Caryophyllene H3C-
-
Journal entries for arrant liabilities and subsequent expenditures. Kingspeed Bikes offers three-year warranties against defects on the sales of its high-end racing bikes. The firm estimates that the...
-
Under its plan of liquidation, Better Corp will distribute all of its assets to its sole shareholder, Barry. The assets have a FMV of $50,000 and an adjusted basis of $14,000. Barry's basis in Corp...
-
Question 2 (20 marks) Ann, Boris, Carrie and Daniel are directors of SpeedRoad Transport Ltd. SpeedRoad is listed in the Australian Security Exchange (ASX) and operates a fleet of 2,000 buses...
-
QUESTIONS: BRIEF a summary of your reaction and lessons learned from the jury deliberation. Answer these questions: Were you able to come to an agreement? Why or why not? What evidence did others...
-
As you continue to explore fundraisingactivities under 45 C.F.R. 164.514(f), I wanted to direct your attention to the following helpful guidance distributed by the Association of American Medical...
-
Jenny and Max are involved in an intimate relationship although Max is married. They decided that they should spice up their sex lives and engage in consensual sexual activities that included...
-
How do we deal with issuance costs and security mispricing costs in our assessment of a projects value?
-
In Exercises 516, find the focus and directrix of the parabola with the given equation. Then graph the parabola. y 2 = 4x
-
Calculate [H 3 O + ] in each aqueous solution at 25 C, and classify each solution as acidic or basic. a. [OH] = 1.1 x 10 M c. [OH = 6.9 10-2 M b. [OH] = 2.9 10 2 M
-
Using Table 15.1 as a guide, write the structural formula for a. Sodium laurate b. Magnesium oleate c. Glyceryl trimyristate d. Glyceryl palmitolauroleate e. Linoleyl stearate f. Methyl arachidate
-
Write equations for the (a) saponification, (b) hydrogenation, and (c) hydrogenolysis of glyceryl trilaurate.
-
Saponification of castor oil gives glycerol and mainly (80 to 90%) ricinoleic acid, also called 12-hydroxyoleic acid. Draw the structure of the main component of castor oil.
-
Having a bit of trouble completing part of the code for my guessing game in java. Basically, I need to add the part of the code that will allow the user to choose how many games they wish to play....
-
Find all the "daffodil numbers" between 100 and 999 and output them. "Daffodil number" refers to a three-digit number, and the cube of each digit is exactly equal to the number itself. For example,...
-
Complete the program so that each cell of array sum contains the sum of the corresponding cells of valA and valB: class Exercise3 { public static void main(String[] args) { int[] valA = {13, -22, 82,...

Study smarter with the SolutionInn App


