Assume that the following compound has the S configuration at its asymmetric carbon. Could this compound be
Question:
Assume that the following compound has the S configuration at its asymmetric carbon.
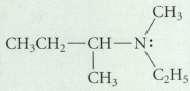
Could this compound be resolved into enantiomers?
Transcribed Image Text:
CHj CH CH2 -CHN: CH CHs
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Each of the rapidly interconverting species in part a ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A classmate tells you that the following compound has the name 3-propylhexane. a. Is he right? If not, what error did he make and what is the correct name? b. How could you redraw the condensed...
-
The following compound has two aromatic rings. Identify which ring is expected to be more reactive toward an electrophilic aromatic substitution reaction.
-
The following compound has two isomers: ClCH==CHCl One isomer has a dipole moment of 0 D, and the other has a dipole moment of 2.95 D. Propose structures for the two isomers that are consistent with...
-
(a) Write the chemical reactions whose equilibrium constants are Kb and Ka for imidazole and imidazole hydrochloride, respectively.
-
Create a chart in which you describe the stages of Piagets theory of cognitive development. For each stage, identify a task that a child could perform.
-
Refer to the SAP Business One SQL query shown in Figure 6.27. The SQL statement is shown in the top of the Queries window.
-
Haar transform (a) For an \(N \times N\) Haar transformation matrix, the Haar basis functions are \[\psi_{k}(t)=\psi_{p q}(t)=\frac{1}{\sqrt{N}} \begin{cases}2^{p / 2}, & (q-1) / 2^{p} \leq t
-
A partnership of attorneys in the St. Louis, Missouri, area has the following balance sheet accounts as of January 1, 2013: Assets .........$320,000 Liabilities ..........$120,000 Athos, capital...
-
Manufacturing costs for Davenport Company during 2018 were as follows: Beginning Finished Goods, January 1 Beginning Raw Materials, January 1 Beginning Work in Process, January 1 Direct Labor for the...
-
Consider a welfare program (such as SNAP) with benefits that decrease as an individuals income increases. Draw the individuals budget constraint with and without the subsidy. (Put hours of work on...
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,2,3,3, -tetramethylbutane
-
Draw a Newman projection of the confirmation of (2S,3R,4S)-2,3,4-hexanetriol (shown above) About the C2{3 bond, with C2 nearest the observer
-
On a recent trip to the Cuyabeno Wildlife Reserve in the Amazon region of Ecuador, Mike took a 100-kilometer trip by speedboat down the Aguarico River from Chiritza to the Flotel Orellana. As Mike...
-
Succession Management Note: Students should read the Case Study prior to starting this assignment. This year, both Dan and Real Seguin are thinking about retiring after approximately50 years of...
-
What can be done to bridge the leadership gender divide and enhance leadership effectiveness as nurse leaders?
-
assume the role of the target audience (i.e., employees) and discuss how implementing communications to address the problems from the post below. Coincidentally, I had a moment at work in this past...
-
Universities are typically organized by departments or colleges such as business, biology, engineering, political science, and so on. Is this an appropriate structure? What aspects of the environment...
-
Organizations have both formal and informal (tacit) structures. How can we predict the effects of calculated and deliberate organizational change on informal networks and structures? How can we...
-
Brianna Ltd is considering these two alternatives to finance its construction of a new $3 million factory: (a) Issue 600 000 shares at the market price of $5 per share. (b) Issue $3 million, 6%...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
The H of hydrogenation is the heat liberated when a compound undergoes catalytic hydrogenation. Consider the H values for hydrogenation of the following three alkenes: 3-methyl -1 -butene, 2126.8 kJ...
-
The heat of formation of (E)-1,3-pentadiene is 75.8 kJ mol 1 (18.1 kcal mol 1 ), and that of 1,4-pentadiene is 106.3 kJ mol 1 (25.4 kcal mol 1 ). (a) Which alkene has the more stable arrangement of...
-
Invoking Hammonds postulate, draw the structure of the reactive intermediate that should most closely resemble the transition state of the rate-limiting step for the hydration of 1-methylcyclohexene....
-
1. A businessman travels between the cities of the following graph. The travel pattern is that he stays in one of the cities for the whole day, and on the next day, he travels to one of the...
-
How do historical legacies of colonialism, imperialism, and systemic injustice continue to shape contemporary power structures, and what steps can be taken to address and redress these historical...
-
Izabella is 20 years old. Eighteen months ago, she started selling some of her old clothing which no longer suited her on peer-to-peer social shopping apps such as Letgo and Depop. She was surprised...

Study smarter with the SolutionInn App


