Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell
Question:
Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell whether the reaction was successful? What differences would you look for in the IR spectra of starting material and product?
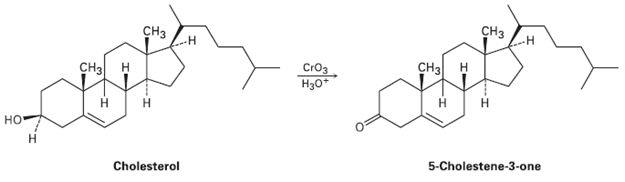
Transcribed Image Text:
ҫHз СHз Сн CHз н CHа н CrOз H30* н но Н 5-Cholestene-3-one Cholesterol -т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The infrared spectra of cholesterol and 5cholestene3one each exhibit a unique absorp...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that you need to prepare 4-methyl-2-pentyne and discover that the only alkynes on hand are acetylene and propyne. You also have available methyl iodide, isopropyl bromide, and 1,...
-
How could you use IR spectroscopy to distinguish between the following pairs of isomers? a. b. c. (CH3CH2)3N and (CH3CH2CH2)2NH 0 CH.CCH,CH, and CH CHCH-CH, CH3 CHCHO and CH CHOCH
-
What features would you look for in a social media outlet? What types of information would you avoid making part of your social media strategy?
-
In September 2020 Kevin sells a drawing for 2,000. He bought the drawing in February 2014 for 50,000 when it was thought (incorrectly) to be by a famous artist. Compute the allowable loss.
-
What are three types of opportunities for sharing that form a sound basis for diversification or vertical integration? Give an example of each from companies you have read about.
-
What statistical procedure can be used to test whether there is a significant difference in the median gray levels between cataractous and normal eyes? Ophthalmology A camera has been developed to...
-
In Example 4.1, \(X\) is the current measured in milliamperes. What is the expected value of power when the resistance is 100 ohms?
-
In October, McEnroe Company reports 21,000 actual direct labor hours, and it incurs $115,000 of manufacturing overhead costs. Standard hours allowed for the work done is 20,000 hours. The...
-
Determine the amount of work (in J) done on an ideal gas as it is heated in an enclosed, thermally isolated cylinder topped with a freely moving piston. The cylinder contains 0.220 mol of the gas and...
-
Aggregate planning: The company has decided to produce a new type of suspension assembly for a new car developed recently. The demands for this specific type of suspension assembly per month for the...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
When the 1HNMR spectrum of an alcohol is run in dimethyl sulfoxide (DMSO) solvent rather than in chloroform, exchange of the OH proton is slow and spinspin splitting is seen between the OH proton...
-
What is an annuity?
-
What happens when a company purchases investments classified as trading securities?
-
The following financial statement information is from five separate companies. Beginning of year Assets Liabilities End of year Assets Liabilities Changes during the year Stock issuances Net income...
-
Liabilities 2010 2011 Assets 2010 2011 Share Capital 100000 120000 Goodwill 15000 13000 Profit/loss a/c 25000 45000 Building 50000 45000 Debenture 50000 75000 Machinery 100000 120000 creditors 10000...
-
How can businesses deploy game theory principles to anticipate and respond to competitive actions, market dynamics, and negotiation scenarios, thereby optimizing their strategic decision-making...
-
How should multinational corporations formulate and execute global expansion strategies that balance standardization and localization, considering diverse cultural, regulatory, and market-specific...
-
In response to the prosecutors questions, a lay witness who is a high school graduate testifies as follows: I measured the defendants skid marks, and I believe he was driving at a speed of at least...
-
A seasonal index may be less than one, equal to one, or greater than one. Explain what each of these values would mean.
-
The following data are given for CCl 4 . Normal melting point, -23 C; normal boiling point, 77 C; density of liquid 1.59 g/mL; fus H = 3.28 kJ mol -1 ; vapor pressure at 25 C, 110 Torr. (a) What...
-
(a) What is the index of hydrogen deficiency of 2-hexene? (b) Of methylcyclopentane? (c) Does the index of hydrogen deficiency reveal anything about the location of the double bond in the chain? (d)...
-
Zingiberene, a fragrant compound isolated from ginger, has the molecular formula C15H24 and is known not to contain any triple bonds. (a) What is the index of hydrogen deficiency of zingiberene? (b)...
-
Carbonyl groups also count for a unit of hydrogen deficiency. What are the indices of hydrogen deficiency for the reactant and for the product in the equation shown at the beginning of Section 4.16...
-
Find the integrating factor for the given 1-order linear non-homogeneous ordinary differential equation. Do not solve the ordinary differential equation. y xdx-xdy-x+dx + dx
-
[Bush] Consider the following snippet of code. (Assume that input strings, including null terminator, will always fit within the size 255 array.) char* to_upper_case(char* original) { char...
-
50. Show that if f(x) = a,x" + a-1x+...+x+ ao, a,..., a-1, and a,, are real numbers and where 0, then f(x) is O(x"). an # Big-O, big-Theta, and big-Omega notation can be extended to functions in more...

Study smarter with the SolutionInn App


