Azulene, a beautiful blue hydrocarbon, is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance
Question:
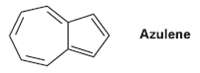
Azulene, a beautiful blue hydrocarbon, is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance form of azulene in addition to thatShown.
Transcribed Image Text:
Azulene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Azulene Azulene is aroma...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Naphthalene is colorless, but its isomer azulene is blue. Which compound has the lower-energy pi electronic transition? naphthalene azulene
-
Azulene is a deep-blue hydrocarbon with resonance energy of 205 kJ mol (49 kcal mol), Azulene has ten pi electrons, so it might be considered as one large aromatic ring. Its electrostatic potential...
-
Draw another resonance form for each of the substituted allylic cations shown in the preceding figure, showing how the positive charge is shared by another carbon atom. In each case, state whether...
-
Data Set 14 in Appendix B lists highway fuel consumption amounts (mi/gal) for cars categorized by size (small, midsize, large). If we use those highway fuel consumption amounts arranged into the...
-
Why is it important for ATS to recruit and train young workers instead of only hiring experienced employees from factories that are closing?
-
Consider the Vasicek Model under risk neutral probability measure: drt = ( rt)dt + dWt (16.3) with r0 = 0.01, = 0.2, = 0.01, = 0.05. Write a Matlab program to simulate a sample path of rt from t...
-
In a random sample of 140 observations of workers on a site, 25 were found to be idle. Construct a \(99 \%\) confidence interval for the true proportion of workers found idle, using the large sample...
-
Suppose a lumber yard has the following data: Accounts receivable, May 31: (.2 * May sales of $360,000) = $72,000 Monthly forecasted sales: June, $437,000; July, $441,000; August, $502,000;...
-
Cost Accounting incl. Full vs. Variable Costing; Budgeting & Financial Planning 1. What is the impact of volume changes in planned capacity on overhead surcharges? How are planned capacity and...
-
Hurzdan, Inc., has a 32-day average collection period and wants to maintain a minimum cash balance of $20 million, which is what the company currently has on hand. The company currently has a...
-
Show the relative energy levels of the seven molecular orbitals of the Cycloheptatrienyl system. Tell which of the seven orbitals are filled in the cation, radical, and anion, and account for the...
-
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic ? system? N. Purine
-
Express the quantity of 422 J in calories.
-
a person has won $500 000. They invest it at 12% compounded semi-annually. They decide to live off the investment, withdrawing an amount every six months. Determine how much money they can withdraw...
-
The following accounts and balances (in thousands) were extracted from the financial statements included in the 2022 10k of ABC, Inc. The company's average tax rate is 30%. Cash $8,000 Cost of goods...
-
15. 16. Define the necessary symbols, and rewrite the argument in symbolic form. 1.If she does not work, then she will not get a paycheck 2.She will get a paycheck Therefore, she does work Complete...
-
Hypothetical comparative condensed balance sheets of Nike, Inc. are presented here. Nike, Inc. Condensed Balance Sheets May 31 ($ in millions) 2025 2024 Assets Current assets $9,880 $8,900 Property,...
-
Suppose that you are undertaking a multiple-choice exam with 20 questions, each of which has five choices of which one is correct. If a student did not study and is randomly guessing. What is the...
-
True or False: The annual percentage rate is the same as the effective annual interest rate.
-
From a medical tourist perspective, compare Shouldice with the traditional hospital in terms of the key factors of competition. Using Table 15-3, why would Shouldice attract patients from outside the...
-
For the reaction N 2 O 4 (g) 2 NO 2 (g), a reaction mixture at a certain temperature initially contains both N 2 O 4 and NO 2 in their standard states, which means that P N2O 4 = 1 atm and P NO 2 =...
-
Give an equation to show the reduction of Tollens reagent by maltose.
-
Does lactose mutarotate? Is it a reducing sugar? Explain. Draw the two anomeric forms of lactose.
-
Trehalose is a nonreducing disaccharide (C12H22O11) isolated from the poisonous mushroom Amanita muscaria. Treatment with an -glucosidase converts trehalose to two molecules of glucose, but no...
-
Frank is 82 years old.His wife died 5 years ago.He died and left the following assets: Principal residence 859,000 (individually owned: ACB 350,000) RRIF $235,000 (Beneficiary adult daughter ACB...
-
Assume that Pope Enterprises held a $10,000, 10 percent, six-month note signed by Mary Drew. On December, 1, 2015, the maturity date, Drew dishonored the note. At this point, Drew owes a total of...
-
5. Consider the shape shown on the following graph and the brute force method of solving the convex hull problem 2 -2 -1 0 a. Label all the points that will be provided to the algorithm as input. You...

Study smarter with the SolutionInn App


