How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic ?
Question:
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic ? system?
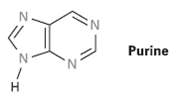
Transcribed Image Text:
N. Purine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
H H N H Purine Purine is a tenxelectron aromatic ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many electrons does each of the following elements have in its outermost electron shell? (a) Magnesium (b) Molybdenum (c) Selenium
-
How many valence electrons does each of the following atoms have? (a) Na (b) Cl (c) Si (d) B (e) Ne (f) N
-
How many electrons does a Ba atom have to lose to have a complete octet in its valence shell?
-
Consider the following velocity distribution curves A and B. a. If the plots represent the velocity distribution of 1.0 L of He(g) at STP versus 1.0 L of Cl2(g) at STP, which plot corresponds to each...
-
Suppose you work in the HR department of a company that wants to hire production workers as independent contractors. What advice would you give management about this idea?
-
Compound S (C8H16) reacts with one mole of bromine to form a compound with molecular formula C8H16Br2. The broadband proton-decoupled 13C spectrum of S is given in Fig. 9.49. Propose a structure for...
-
Barry Hannah, CPA, plans to use attributes estimation to help assess control risk for the Oxford Company's control procedures over credit sales transactions. Hannah has begun to outline main steps in...
-
Renkas Heaters selected data for October 2011 are presented here (in millions): Direct materials inventory 10/1/2011 $ 105 Direct materials purchased 365 Direct materials used 385 Total manufacturing...
-
On 1 August the balance of the Accounts receivable control account for Solid Rental Company was $10 500. The customers' subsidiary ledger contained account balances as follows: Anton $3 000 Carmine...
-
Portfolio return and standard deviation David Choo is thinking of building an investment portfolio containing two stocksH and G. He has assigned 40% of his portfolio to stock H and the remaining 60%...
-
Azulene, a beautiful blue hydrocarbon, is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance form of azulene in addition to thatShown. Azulene
-
Give IUPAC names for the following substances (red = O, blue =N): (a) (b)
-
What is the probability of randomly selecting a diamond from a standard 52-card deck?
-
Frenz, Frezell and Fatima, recently passed the bar exam. They decided to form F3 Legal Services contributing P48,000, P80,000 and P120,000 respectively. During the year, frezell and Fatima withdrew...
-
The constant force F = 7i + 4j +-3k , N acts on a particle during a displacement from position in coordinate origin to position r = 4i + 3j +2k , m. Determine the total work done on the particle.
-
A double-slit experiment is performed using a slit separation of 0.12 mm with a screen placed 80.0 cm away. There are 18 mm between the first and seventh nodal lines. What wavelength of light was...
-
A 10kg Ice Skater is moving at 6 m/s and is headed towards a stationery 4kg snowman. After the two collide, the ice skater has 0 m/s of velocity and the snowman moves forward. What is the velocity of...
-
A certain department produced 16,000 units of a product that had a standard materials cost of two pounds at $1.70 per pound and a standard labor cost of two hours per unit at $9.40 per hour. Actual...
-
The first-order di ffraction of x rays from two crystals with simple cubic structure is measured. The first-order di ffrac tion from crystal A occurs at an angle of20. The first-order diffraction of...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
Consider the reaction of A to form B: A reaction mixture at 298 K initially contains [A] = 0.50 M. What is the concentration of B when the reaction reaches equilibrium? 2 A(g) = B(g) Kc = 1.8 x 105...
-
Raffinose is a trisaccharide (C18H32O16) isolated from cottonseed meal. Raffinose does not reduce Tollens reagent, and it does not mutarotate. Complete hydrolysis of raffinose gives D-glucose,...
-
Cellulose is converted to cellulose acetate by treatment with acetic anhydride and pyridine. Cellulose acetate is soluble in common organic solvents, and it is easily dissolved and spun into fibers....
-
Cytosine, uracil, and guanine have tautomeric forms with aromatic hydroxyl groups. Draw these tautomeric forms.
-
Water flows through a pipe section that tapers from an internal diameter of 200 mm down to 100 mm over a distance of 1 m. Determine the pressure drop over the section for a flow of 0.06 m3s-1. The...
-
Calculate the binding energy per nucleon for Be, 59 Co, 208 pb, and 195 pt. (For the atomic masses, see this table. Enter your answers to at least two decimal places.) (a) Be MeV/nucleon 59 Co...
-
A.Final ProjectTeamProjectCustom Schema: 200 points Extra Credit Group of 3 people : Designer and Developer ( ERD, tables, data ) Team Leader/Power User ( Business Questions/Queries, Real Life...

Study smarter with the SolutionInn App


