Base treatment of the following ?, ?-unsaturated carbonyl compound yields an anion by removal of H from
Question:
Base treatment of the following ?, ?-unsaturated carbonyl compound yields an anion by removal of H from the y carbon. Why is hydrogen?s on the y carbon atom acidic?
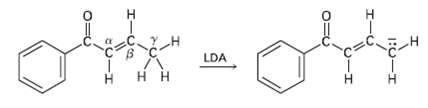
Transcribed Image Text:
LDA 1:0-I I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
Enolization at the y position produc...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Treatment of the following alkene with a peroxyacid yields an epoxide different from that obtained by reaction with aqueous Br2 followed by base treatment. Propose structures for the two epoxides,...
-
Treatment of the following epoxide with aqueous acid produces a carbocation intermediate that reacts with water to give a diol product. Show the structure of the carbocation, and propose a mechanism...
-
An carbonyl compound can be prepared by a reaction known as a selenenylation-oxidation reaction. A selenoxide is formed as an intermediate. Propose a mechanism for the reaction. 1. LDA/THF 2. CgHSeBr...
-
On January 1, 2012, Wilmes Floral supplies borrowed $2,413 from Bower Financial Services. Wilmes Floral Supplies gave Bower a $2,500 note with a maturity date of December 31, 2013. The note specified...
-
What are some tips for business writers wishing to apologize effectively?
-
An experiment was conducted to determine whether either firing temperature or furnace position affects the baked density of a carbon anode. The data are as follows: (a) State the hypotheses of...
-
What kind of responsibility (attribute or operation) are personal data? Explain.
-
Marc and Michelle are married and earned salaries this year (2009) of $64,000 and $12,000, respectively. In addition to their salaries, they received interest of $350 from municipal bonds and $500...
-
ANSWER THIS QUESTION ACCURATELY. DO NOT ATTEMPT IF YOU DON'T KNOW WHAT YOU'RE DOING AND DO NOT USE AI/CHATGPT. I will find out. Link to reference textbook:...
-
Steve Reese is a well-known interior designer in Fort Worth, Texas. He wants to start his own business and convinces Rob ODonnell, a local merchant, to contribute the capital to form a partnership....
-
One way to determine the number of acidic hydrogens in a molecule is to treat the compound with NaOD in D20, isolate the product, and determine its molecular weight by mass spectrometry. For example,...
-
Treatment of 1-phenyl-2-propenone with a strong base such as LDA does not yield an anion, even though it contains a hydrogen on the carbon atom next to the carbonyl group.Explain....
-
The data show the number of U.S. space launches for the 10-year periods from 1960 to 2009. Construct a time series graph for the data and analyze the graph. Year Launches 614 247 199 60-69 70-79...
-
Solve the following differential equation. (2xsiny-y+x)dx+ (x'cosy - 3xy2-2y)dy = 0
-
Suppose the current spot rates are $.135/MXN ask and $.133/MXN bid, combined with current spot rates of .775/$ ask and .765/$ bid. What are the /MXN bid and /MXN ask prices? (to 4 decimal places)...
-
In assessing data that fell into two phenotypic classes, a geneticist observed values of 800: 680. She decided to perform a chi-square analysis by using the following null hypothesis: The data fit...
-
How do individual differences and diversity contribute to the dynamics of team performance in a multicultural organization? Explain
-
Analyzing and Reporting Financial Statement Effects of Bond Transactions On January 1 of the current year, Banek Inc. issued $ 7 7 0 , 0 0 0 of 8 % , nine year bonds for $ 6 7 9 , 9 9 0 , which...
-
A Temple University mathematics professor used these data to show that most Americans have an exaggerated fear of terrorists : Without some feel for probability, car accidents appear to be a...
-
Explain the Hawthorne effect.
-
Determine the hybridization about carbon in H 2 CO. a) sp b) sp 2 c) sp 3 d) sp 3 d
-
Two general mechanisms (or various versions of them) for alkene metathesis were originally considered. In the first (pair wise) mechanism, shown in Fig. P18.81, the catalyst brings about cyclobutane...
-
Citaprolam is used as an antidepressant. N(CH,)2 Citaprolam
-
What is dn for each of the following complexes? Pd(PPh3)4
-
s Machine-hours required to support estimated production Fixed manufacturing overhead cost Variable manufacturing overhead cost per machine-hour Required: 1. Compute the plantwide predetermined...
-
How do visionary leaders navigate the complexities of organizational dynamics to cultivate a culture of innovation and adaptability ? Explain
-
Finnish Corporation manufactures custom molds for use in the extrusion industry. The company allocates manufacturing overhead based on machine hours. Selected data for costs incurred for Job 532 are...

Study smarter with the SolutionInn App


