Treatment of 1-phenyl-2-propenone with a strong base such as LDA does not yield an anion, even though
Question:
Treatment of 1-phenyl-2-propenone with a strong base such as LDA does not yield an anion, even though it contains a hydrogen on the carbon atom next to the carbonyl group.Explain.
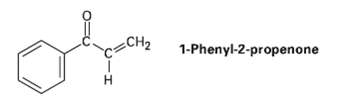
Transcribed Image Text:
1-Phenyl-2-propenone CH2 H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The illustrated compound 1phenyl2propenone ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Treating with PGA2 strong base such as sodium tert-butoxide followed by addition of acid converts it to PGC2. Propose a mechanism for this reaction.
-
In the dehydrohalogenation of alkyl halides, a strong base such as tert-butoxide usually gives the best results via the E2 mechanism. (a) Explain why a strong base such as tert-butoxide cannot...
-
(S)-1-Bromo-1,2-diphenylethane reacts with a strong base to produce cis-stilbene and trans-stilbene: a) This reaction is stereo-selective, and the major product is trans-stilbene. Explain why the...
-
The balance sheet of Lamont Bros. follow: (a) What portions of Lamont's assets were provided by debt, contributed capital, and earned capital? Reduce contributed capital by the cost of the treasury...
-
When is the indirect strategy in communicating bad news preferable?
-
An experiment has run a single replicate of a 2 4 design and calculated the following factor effects: (a) Construct a normal probability plot of the effects. (b) Identify a tentative model, based on...
-
How do you mine use cases to find classes?
-
The Hit-and-Miss Manufacturing Company produces items that have a probability p of being defective. These items are produced in lots of 150. Past experience indicates that p for an entire lot is...
-
The trial balance columns of the worksheet for Carla Vista Roofing at March 31, 2022, are as follows. Carla Vista Roofing Worksheet For the Month Ended March 31, 2022 Trial Balance Account Titles...
-
Q1\ Find dy dx , if y=t-3, x= 3t (5 marks)
-
Base treatment of the following ?, ?-unsaturated carbonyl compound yields an anion by removal of H from the y carbon. Why is hydrogen?s on the y carbon atom acidic? LDA 1:0-I I.
-
When optically active (R)-2-rnethylcyclohexanone is treated with either aqueous base or acid, racemization occurs. Explain.
-
Show that where T k is the kth triangular number. (See Question 4 in Exercises 7.2.3.) Data from Question 4 in Exercises 7.2.3. Triangular numbers (T n ) are defined by the number of dots that occur...
-
Mimosa Inc. has total assets of $6,321,600 at the beginning of the year and $4,896,500 at the end of the year. If its net income during the year is $499,500, what is Mimosa's return on assets for the...
-
How do endocrine disorders, such as diabetes mellitus, hyperthyroidism, and hypopituitarism, arise from dysregulation of hormone synthesis, secretion, or receptor signaling, and what are the...
-
What are the implications of social learning theory on the development of organizational culture and norms? Explain
-
BRIEFLY DESCRIBE an essay for THESE THREE THINGS (TOPICS) YOU WILL ADDRESS. Define the goals of business writing, summarize the 3-x-3 writing process, and explain how it guides a writer. Analyze the...
-
Not every case subject to review by a court is a case involving constitutional law. Why? How can a court determine when a case involves constitutional law? How can a legal researcher make that...
-
In an interview for a banking job, a student was asked to calculate the following: A can contains 20 coins, 19 of which are ordinary coins and 1 of which has heads on both sides. A coin is randomly...
-
Evaluate the integral, if it exists. Jo y(y + 1) dy
-
What is the molecular geometry about nitrogen in CH 3 NH 2 ? (a) Trigonal planar (b) Tetrahedral (c) Trigonal pyramidal (d) Bent
-
Within set, rank the compounds in order of increasing rates of their SN2 reactions. Explain your reasoning. 1-bromocyclohexene, bromocyclohexane, 1-(bromomethyl)cyclohexene
-
How many CO ligands would be accommodated by Fe(O) if we assume that the resulting complex follows the 18-electron rule?
-
Give the structure for each of the following compounds. (a) Valerophenone (b) -chlorobutyraldehyde (c) 3-hydroxy-2-butanone (d) 3-cyclohexenone
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
The York City Hospital has just acquired new equipment. The equipment cost $ 4 , 2 5 0 , 0 0 0 , and the organization spent $ 1 3 5 , 0 0 0 on upgrading the physical plant to the new equipment will...
-
Calculate the following showing all the necessary steps: Note: CPP rate to be used in the calculation is 5.95%. Note: Don't forget to deduct pay period exemption\ \ Tanya earns $25.00 per hour. This...

Study smarter with the SolutionInn App


