Question: Both cis and trans alkenes can be formed from this compound y anti elimination. Draw a Newman projection of the conformation required to form each
Both cis and trans alkenes can be formed from this compound y anti elimination. Draw a Newman projection of the conformation required to form each of these products and, on the basis of these projections predict which of these products would be formed in largeramounts?
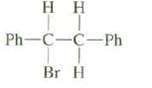
Ph-C-C-Ph Br H
Step by Step Solution
3.51 Rating (161 Votes )
There are 3 Steps involved in it
The conformation that produces the tra... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-E-R (4).docx
120 KBs Word File


