Calculate formal charges for the non-hydrogen atoms in the following molecules: (a) Diazomethane, (b) Acetonitrile oxide, (c)
Question:
Calculate formal charges for the non-hydrogen atoms in the following molecules:
(a) Diazomethane,
(b) Acetonitrile oxide,
(c) Methylisocyanide,
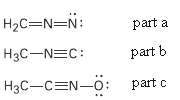
Transcribed Image Text:
H2C=N=N: part a H3C-NEC: part b H3C-CEN-o: part c
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Strategy To find the formal charge of an atom in a molecule follow these two steps 1 Draw an e...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH CHCH2CHCH2CH Gs (c) (a) (b) CH2H2CH CHCCH2CCH CH
-
A few hydrogen and oxygen molecules are introduced into a container in the quantities depicted in the following drawing. The gases are then ignited by a spark, causing them to react and form H2O. a....
-
In the following four models, C atoms are black, H atoms are light blue, O atoms are red, and N atoms are dark blue: a. Write the molecular formula of each molecule. b. Write the condensed structural...
-
True Or False Death benefits are used to compensate the deceaseds family for pain and suffering.
-
Near the end of 2016, Byron realizes that he has a net short-term capital loss of $13,000 for the year. Byron has taxable income (not including the loss) of $123,000 and is single. He owns numerous...
-
Write a mass balance for a solution of Fe2(SO4)3 if the species are Fe3+ , Fe(OH)2 , Fe(OH)+2 , Fe2(OH)4+2 , FeSO+4 , , and SO2-4 . andHSO2-
-
Air flows steadily between two sections in a duct. At section (1), the temperature and pressure are \(T_{1}=80^{\circ} \mathrm{C}, p_{1}=301 \mathrm{kPa}(\mathrm{abs})\), and at section (2), the...
-
The Morning Brew Coffee Shop sells Regular, Cappuccino, and Vienna blends of coffee. The shop's current daily labor cost is $320, the equipment cost is $125, and the overhead cost is $225. Daily...
-
What steps are involved when monitoring performance? Give three (3) good key performance indicators in relation to your role in the organisation. 9. What are the different ways an organisation can...
-
Cranston Berries sells three types of berries: strawberries, blueberries, and raspberries. Sales have been booming this year and are expected to increase next year. The sales manager wants a program...
-
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane :O:
-
Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphatedianion. 2- :0: H-C-0-P-0: Methyl phosphate :0:
-
Find the function r that satisfies the given condition. r' (t) = (1, 2t, 3t); r(1) =(4,3,-5)
-
DRAW THE GRAPHS OR AT LEAST EXPLAIN WHAT CURVE SHIFTS AND WHY China joined WTO in December 2001. As a result, the Chinese current account has been increasing and the U.S. current account has been...
-
Smith had no stock transactions in 2 0 2 1 , so the change in stockholders' equity for 2 0 2 1 was due to net income and dividends. If dividends were $ 5 0 , 0 0 0 , how much was Smith's net income...
-
a. The information given below pertains to three companies which recently paid exactly the same dividends of RM2.00 per share. However, the future annual growth rate in dividends, g, for each firm is...
-
Peter and Doug are neighbors who hate one another. One day, Doug is nailing some boards together on the common sidewalk thathe shares with Peter. In a classic slapstick comedy move, Doug picks up a...
-
3. 4. your What will future staffing needs be? Use the data at the end of the case to determine answer. Discuss the approach you take to solve the problem and provide your work in an appendix. Be...
-
Based on Exhibit 1 and Exhibit 2, the RedGrass Funds 2017 investmentmanagement fee is: A. 0.40%. B. 1.00%. C. 2.60%. The Tree Fallers Endowment plans to allocate part of its portfolio to alternative...
-
What are the three kinds of research types? Explain each type.
-
a. Quantitatively explain why ice skates slide along the surface of ice. Can it get too cold to ice skate? b. Is it possible to ice skate on other materials, such as frozen carbon dioxide? c. What is...
-
Explain why the pKas of compounds near the middle of Table 4.2 are often listed with two figures to the right of the decimal place (that is, for NH4+ the pKa = 9.24), whereas those at the beginning...
-
For each pair of compounds, explain which is the stronger acid?
-
Explain why the compound on the left is a stronger acid than the compound on the right.
-
A manufacturer reports finished goods inventory of $820 on June 1 and $1,130 on June 30. Cost of goods manufactured for June is $5,460. What is cost of goods sold for June? Cost of Goods Sold Cost of...
-
Stacey's Piano Rebuilding Company has been operating for one year. At the start of the second year, its income statement accounts had zero balances and its balance sheet account balances were as...
-
MyBnB started a home rental company on January 1. As of November 30, MyBnB reported the following balances. The company does not yet have a balance in Retained Earnings because this is its first year...

Study smarter with the SolutionInn App


