Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH
Question:
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary:
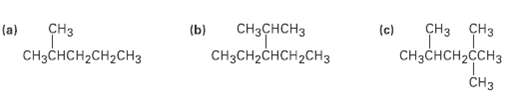
Transcribed Image Text:
ҫHз ҫHз CHз CнзснсHз CHзCH2CHCH2CHз Сна Сна Gнs (c) (a) (b) CнзснсH2сH2CHз CHзCнCH2CCHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
a b PIP CH3CHCH3 CHCHCHCHCH3 ...View the full answer

Answered By

Gabriela Rosalía Castro
I have worked with very different types of students, from little kids to bussines men and women. I have thaught at universities, schools, but mostly in private sessions for specialized purpuses. Sometimes I tutored kids that needed help with their classes at school, some others were high school or college students that needed to prepare for an exam to study abroud. Currently I'm teaching bussiness English for people in bussiness positions that want to improve their skills, and preparing and ex-student to pass a standarized test to study in the UK.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the carbon atoms expected to be labeled with 14C when each of the following substances is biosynthesized from acetate enriched with 14C in its methyl group: (a) (b) (c) (d) Palmitic acid CH3...
-
Identify the carbon atoms expected to be labeled with 14C when each of the following substances is biosynthesized from acetate enriched with 14C in its methyl group:
-
Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2 "CH2 - H Hydroxyacetone Crotyl alcohol
-
In a bank reconciliation, deposits in transit are: (a) deducted from the book balance. (b) added to the book balance. (c) added to the bank balance. (d) deducted from the bank balance.
-
In what ways do you think the subject matter in this strategic management-business policy course will differ from that of previous courses you have taken?
-
Khaled Al-Zubari, an executive recruiter, is a member of the board of directors of Mantilla Corporation. At a recent board meeting, called to discuss the financial plan for 20X4, Mr Al-Zubari...
-
You have fit a linear regression model with three predictors to a sample of 50 observations. The total sum of squares is 150 and the regression sum of squares is 120 . The estimate of the error...
-
Barts Company has prepared the PP&E and depreciation schedule shown in Exhibit 8.46.1 below. The following information is available (assume the beginning balance has been audited): The land was...
-
The expected yield on a 2 year security is 7.8%. If the yield on a t-bill maturing in 1 year is 7.2%, what is the yield on a 1 year t-bill maturing 1 year from now?
-
David Sinclair operates a widget shop. The shop specializes in a selection of various widget products. You have been hired as manager. Your duties include maintaining the store?s financial records....
-
Draw the eight 5-carbon alkyl groups (pentyl isomers).
-
Identify the hydrogen atoms on the compounds shown in Problem 3.8 as primary, secondary, or tertiary.
-
The probability of success on a single trial of a binomial experiment is known to be .The random variable x, number of successes, has a mean value of 80. Find the number of trials involved in this...
-
Directors and officers who act in breach of their fiduciary duties owed to the corporation and its shareholders can be personally liable for their actions. Are there any circumstances under which an...
-
Why would the diversity of languages spoken by persons within the healthcare workforce create teamwork problems among healthcare staff? What language etiquette could ease interpersonal and...
-
A 6.5 kg object undergoes an acceleration of 3.6 m/s? What is the magnitude of the resultant force acting on it? If this same force is applied to a 3.9 kg object, what acceleration is produced?
-
What are the psychological underpinnings of social cohesion and collective identity, and how do factors such as intergroup conflict, prejudice, and social exclusion impact the fabric of society?
-
The coefficient of static friction between the m = 2.75-kg crate and the 35.0 incline of the figure below is 0.325. What minimum force F must be applied to the crate perpendicular to the incline to...
-
Should MLB adopt a system of promotion and relegation?
-
Define cultural intelligence. Cite the books or journal articles you found in Capella's library. Explain why cultural intelligence is important for HR practitioners and other organizational managers.
-
Estimate how much heat is absorbed when 1.00 g of Instant Car Kooler vaporizes. Comment on the effectiveness of this spray in cooling the interior of a car. Assume the spray is 10% C 2 H 5 OH(aq) by...
-
(a) Outline a likely mechanism for the solvomercuration step of the ether synthesis just shown. (b) Show how you would use solvomercuration-demercuration to prepare tertbutyl methyl ether. (c) Why...
-
Specify the alkene needed for synthesis of each of the following alkylboranes by hydroboration: (a) (b) (c) (d) Show the stereochemistry involved in the hydroboration of 1-methylcyclohexene.
-
Treating a hindered alkene such as 2-methyl-2-butene with BH3: THF leads to the for- mation of a dialkylborane instead of a trialkylborane. When 2 mol of 2-methyl-2-butene is added to 1 mol of BH3,...
-
Record the following transactions for Tri - State Corporation on the dates indicated. 1 . On March 3 1 , 2 0 2 0 , Tri - State Corporation discovered that Depreciation Expense on equipment for the...
-
William has decided to increase his sales price to $ 2 6 to offset the supplier's price increase. He believes that the increase will result in a 5 % reduction from last year's sales volume. What is...
-
The same scenarios exist as before, with labor costing either $ 1 3 / lawn or $ 2 5 / lawn . Assume that your mowing business will mow 3 5 0 lawns per month rather than 2 0 0 lawns per month. Also...

Study smarter with the SolutionInn App


