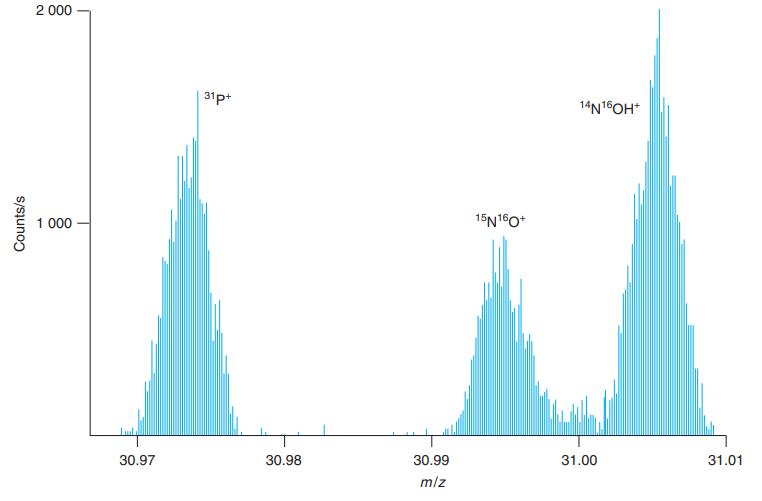
Calculate the theoretical masses of the species in Figure 21-9 and compare your answers with the values
Question:
Calculate the theoretical masses of the species in Figure 21-9 and compare your answers with the values observed in the figure.
Figure 21-9
Transcribed Image Text:
2 000 31p+ 14N16OH* 1 000 15N160* 30.97 30.98 30.99 31.00 31.01 m/z Counts/s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
31P 31P e 30973 760000 55 30973 21 observed 309735 To measure mz I enlar...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Calculate the theoretical air-fuel ratio on a mass and mole basis for the combustion of ethanol, C2H5OH.
-
Calculate the theoretical air-fuel ratio on a mass and mole basis for the combustion of ethanol, C2H5OH.
-
Calculate the theoretical density of -Sn. Assume the diamond cubic structure and obtain the atomic radius information from Appendix B.
-
What implications might the difference in initial training provision between the USA and Germany have for the organisation of work and the character of relationships between managers and...
-
Terando Co. began operations on July 1. It uses a perpetual inventory system. During July, the company had the following purchases and sales. Instructions (a) Determine the ending inventory under a...
-
Pamela Albright is the manager of the audit of Stanton Enterprises, a public company that manufactures formed steel subassemblies for other manufacturers. Albright is planning the 2011 audit and is...
-
What are the primary categories of a risk?
-
Statistics for three stocks, A, B, and C, are shown in the following tables. Only on the basis of the information provided in the tables, and given a choice between a portfolio made up of equal...
-
Granada Inc. applies overhead to production at a predetermined rate of 0.79 based on direct labor cost. Job No. 250, the only job still in process at the end of September, has been charged with...
-
Volcano Potato Company (VPC) grows potatoes, processes them, and then sells three potato products: fresh potatoes, frozen french fried potatoes, and frozen hash ball potatoes (shredded and then...
-
The mass of a fragment ion in a high-resolution spectrum is 83.086 5 Da. Which composition, C 5 H 7 O + or C6H + 1 1, better atches the observed mass?
-
Consider the extraction of Mn+ from aqueous solution into organic solution by reaction with protonated ligand, HL: Rewrite Equation 22-13 in terms of Kextraction and express Kextraction in terms of...
-
Graph the solution set of the inequality, where x is an integer, on the number line. 1 > -x > -6
-
The Chicago Mercantile Exchange houses the fed funds futures market as well as a number of other interest rate options and futures. Go to http://www.cmegroup.com/...
-
Go to www.sec.gov/ and click on Press Releases. a. Summarize the major types of issues that the SEC addresses in these press releases. b. Review the past three months of releases, and count how many...
-
Go to http://www.soxlaw.com/. This site tracks issues and news related to the Sarbanes-Oxley Act. a. Summarize in two or three sentences the primary reason for the passage of the Sarbanes-Oxley Act....
-
You have been out of school and gainfully employed for 5 years. You have three alternatives available for investment with your own money. Each has some element of risk, although some are safer than...
-
The Federal Reserve has a listing of the nonconventional policy tools used for liquidity provision at https://www.federalreserve.gov/monetarypolicy/ expiredtools.htm. Of those tools, which one was...
-
Showing the details of your work, find L(f) if f(t) equals: 1/2t 2 sin t
-
What types of questions can be answered by analyzing financial statements?
-
A 50.0-mL solution containing Ni 2+ and Zn 2+ was treated with 25.0 mL of 0.045 2 M EDTA to bind all the metal. The excess unreacted EDTA required 12.4 mL of 0.012 3 M Mg 2+ for complete reaction. An...
-
Considering just acid-base chemistry, not ion pairing and not activity coefficients, use the systematic treatment of equilibrium to find the pH and concentrations of species in 1.00 L of solution...
-
What is the difference between E and E o for a redox reaction? Which one runs down to 0 when the complete cell comes to equilibrium?
-
A farmer wonders if his crops grow better in sun or in shade. He measures the amount of fruit gathered from a sample of 50 trees growing in full sun and from a sample of 50 trees growing in mostly...
-
Consider the diagram. Determine the length of AC to the nearest cm. Determine the length of DC to the nearest cm. 28 38 A 224 cm 789 B
-
Use this picture to answer the following questions. 10 18 16. 3 AB=10 AC=16 AD=22 AE=15 BC-17 BD=18 15 BE=25 CD=26 CE=8 DE=11 Find the Nearest Neighbor Circuit for each vertex. Also give the weight...

Study smarter with the SolutionInn App


