Cinnamaldehyde, the aromatic constitution of cinnamon oil, can be synthesized by a mixed aldol condensation. Show the
Question:
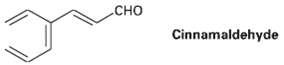
Cinnamaldehyde, the aromatic constitution of cinnamon oil, can be synthesized by a mixed aldol condensation. Show the starting materials you would use, and write thereaction.
Transcribed Image Text:
сно CHO Cinnamaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
CHO CH3CHO 1 NaOH EtOH 2 heat ...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following compounds can be synthesized by aldol condensations, followed by further reactions. (In each case, work backward from the target molecule to an aldol product, and show what compounds...
-
Epoxides can be synthesized by treating halohydrins with aqueous base. Propose a mecha-nism for reactions (a) and (b), and explain why no epoxide formation is observed in (c). (a) (b) (c) CI NaOH H20...
-
Vicinal halo alcohols (halohydrins) can be synthesized by treating epoxides with HX. (a) Show how you would use this method to synthesize 2-chlorocyclopentanol from cyclopentene. (b) Would you expect...
-
4. Jerry intends to use the money from his loan (and his personal savings if necessary) to make an investment in his friend Elaines business. In return, Elaine has predicted the following returns on...
-
Suppose you were granted a "risky job" of the type studied in this chapter. The job pays $40,000 with probability 1/2, and $160,000 with probability 1/2. What is your certainty equivalent for this...
-
Figure E16.26 shows cascade temperature control of a polymerization reactor, which uses feed heat exchange to adjust the reactor temperature. Using the instrumentation diagram, explain how this...
-
Can a patient consent to a procedure and then withdraw it? Discuss your answer.
-
2015, Aron Company purchases 100% of the common stock of Shield Company for $450,000 cash. On this date, any excess of cost over book value is attributed to accounts with fair values that differ from...
-
Basic Summary Statistics Problem: Percent of State Expenditures for Roads and Bridges that comes from user fees. An article noted that owners of EV vehicles do not pay gas taxes that are often...
-
A social scientist has noticed that people seem to be spending a lot of nonwork hours on computers and wants to determine if this may, in some way, be associated with social relationship satisfaction...
-
How can you account for the fact that 2, 2, 6-trimethylcyclohexanone yields no detectable aldol product even though it has an acidic hydrogen?
-
The bicycle ketone shown below does not undergo aldol self-condensation even though it has two ? hydrogen atoms. Explain.
-
Suppose that firms in the ocean shipping business can sell their decommissioned ships for scrap steel (to be recycled). If the price of scrap steel rises, how would the supply function of a firm in...
-
Hardball Stores' beta is 2.27. The market risk premium is 7%. The risk-free rate is 1.7%. Find Hardball's risk premium.
-
Build the yield curves based on spot rates and forward rates. Comment on the slopes. What do they tell you about the behavior of interest rates in the future?
-
Question 48 (Mandatory) (1 point) _____ involves reducing the impact of a risk event by reducing the probability of its occurrence. Question 48 options: a) Risk acceptance b) Risk avoidance c) Risk...
-
Associate the following descriptions with their appropriate financial terminology. Answer instructions The procedure comprises planning and projecting the business's financial assets Financial...
-
All of the following items would qualify as hedging instruments, except Multiple choice question. call options. interest rate swaps. insurance contracts. futures contracts.
-
John has taxable income of $30,000. William has taxable income of $60,000. Determine their 2017 income taxes if they are both single individuals and claim the standard deduction. Compare their...
-
For what reason might an exporter use standard international trade documentation (letter of credit, draft, order bill of lading) on an intrafirm export to its parent or sister subsidiary?
-
In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF 2 , however, oxygen is the central atom. Use formal charges to explain why. Formal charge = number of valence electrons...
-
Give the expected product(s) of E2 elimination for each reaction. (Hint: Use models!) (a) (b) CH, H NaOCH one product HC H NaOCH two products
-
Determine the number of elements of unsaturation in the molecular formula C4H6. Give all nine possible structures having this formula. Remember that A double bond = one element of unsaturation A ring...
-
The preceding example shows meso-1,2-dibromo-1,2-diphenylethane reacting with iodide ion to give trans-stilbene. Show how the other diastereomer of the starting material gives a different...
-
123 Anna purchased 100 shares of Delta stock on February 1, Year 2, for $46 per share, and 5 received a two-for-one stock split on December 31, Year 2. Anna sold all the shares of Delta stock on...
-
work i Saved QS 17-10 (Algo) Computing activity rates for activity-based costing LO P3 A company sells two types of products: standard and deluxe. It prepares the following analysis showing budgeted...
-
! Required information [The following information applies to the questions displayed below.] Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It...

Study smarter with the SolutionInn App


