In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF
Question:
In N2O, nitrogen is the central atom, and the oxygen atom is terminal. In OF2, however, oxygen is the central atom. Use formal charges to explain why.
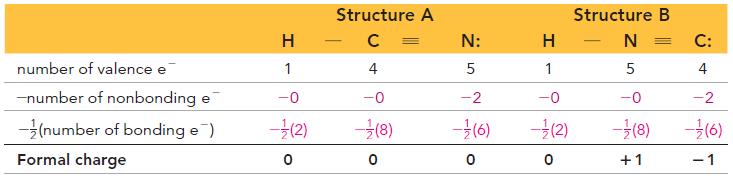
Transcribed Image Text:
Formal charge = number of valence electrons - (number of nonbonding electrons + number of bonding electrons) So the formal charge of hydrogen in HF is 0: Formal charge = 1 - [0+ (2)]=0 Number of valence electrons for H Number of electrons that H "owns" in the Lewis structure Similarly, we calculate the formal charge of fluorine in HF as 0: Formal charge = 7 - [6+ (2)]=0 Number of valence electrons for F Number of electrons that F "owns" in the Lewis structure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The question is asking us to use formal charges to explain why nitrogen is the central atom in N 2 O while oxygen is the central atom in OF 2 Formal c...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a GUI application that records items of stock for a business. A barcode, description, stock level and price are recorded for each item. New items must be able to be added, and existing items...
-
Oxidation of the cyanide ion produces the stable cyanate ion (OCN2). The fulminate ion (CNO2), on the other hand, is very unstable. Fulminate salts explode when struck; Hg(CNO)2 is used in blasting...
-
Use the LE model to describe the bonding in H2CO and C2H2. Carbon is the central atom in H2CO, and C2H2 exists as HCCH.
-
L-keys, such as the one shown in the figure below, are commonly used to insert screws and bolts in medical implants. This insertion process can be simplified into the loading scenario shown in the...
-
What is the difference between sorting records and indexing records in a database?
-
Cylinder 1 of diameter 200mm and cylinder 2 of diameter 300 mm are placed in a trough as shown in fig. 3. If cylinder 1 weighs 800 N and cylinder B weight 1200 N, determine the reactions developed at...
-
The Anchor Glass Container Corporation and its parent company, Consumers Packaging, Inc. (CPI), entered into a series of agreements with Encore Glass, Inc., to supply glass containers of a specific...
-
Greg Davis, a business major at the University of South Carolina (USC), has opened Six Points Saco (SPS), a specialty subs-taco restaurant, at the rim of the USC campus. SPS has grown in popularity...
-
Create a table in excel showing net present value calculation for the following scenario. You will have a column with time period, future value, and present value included. You can anticipate that...
-
How important is the resonance structure shown here to the overall structure of carbon dioxide? Explain. :0=C:
-
Use formal charge to identify the better Lewis structure. H H-C= H H=C
-
Beginning three months from now, you want to be able to withdraw $3,000 each quarter from your bank account to cover college expenses over the next four years. If the account pays .57 percent...
-
Find one example of institutional transitions from developed economies and one example from emerging economies. What are their similarities and differences?
-
Assume that the per-worker production function is y t = 2k t 0.5 . The saving and depreciation rates are estimated at 0.2 and 0.04, respectively. a) Calculate the capital-labor ratio steady state for...
-
Consider the world economy and comment on the effect of the Industrial Revolution on the world growth rate of output per person, according to the assumptions of the Romer model.
-
Compare and contrast the five forces affecting the cruise industry, fashion industry, airline industry, and automobile industry (1) on a worldwide basis (2) in your country. Which industry holds more...
-
What determines the desired amounts of national saving and investment? What relationship between desired saving and desired investment is required for goods market equilibrium, and how is this...
-
If countries are so keen to reduce the barriers to trade, why do many countries frequently attempt to erect barriers?
-
Write the general quadratic equation y2 - 8y - 4x + 28 = 0 in standard form. Determine the vertex, focus, and directrix of the parabola defined by this equation. Sketch a graph.
-
Why is mercury a convenient fluid to use in a barometer?
-
If water were to be used instead of mercury in a barometer, how high would the water column be?
-
What is the barometric pressure reading in inches of mercury corresponding to 14.696 psia?
-
Although achieving horizontal and vertical fits are important for high-performance work systems, what do you consider more critical and why?
-
What are the biggest issues that need to be addressed? Given the current situation, how would you proceed in restoring motivation? Be sure to relate theories and strategies leading to certain best...
-
Contrast the differences between straight piecework, differential piece rate, and standard hour plans. Explain where each plan might best be used. What are the reasons behind the different payment...

Study smarter with the SolutionInn App


