Compound A, C 4 H 8 O 3 , has infrared absorptions at 1710 and 2500 to
Question:
Compound A, C4H8O3, has infrared absorptions at 1710 and 2500 to 3100 cm?1 and has the 1H NMR spectrum shown. Propose a structure for A.
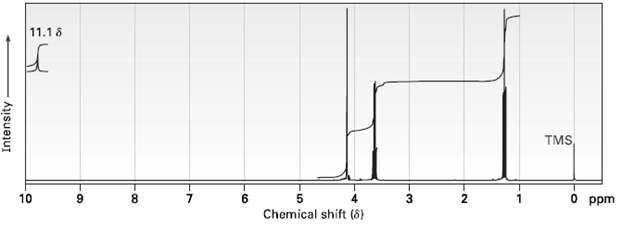
Transcribed Image Text:
11.18 TMS 10 O ppm 8. 6. 3. Chemical shift (6) Intensity -00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
The compound has one degree of ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
-
The 1H NMR spectrum shown is that of 3-methyl-3-buten-l-ol. Assign all the observed resonance peaks to specific protons, and account for the splittingpatterns. TMS H H2H20 10 8. O ppm Chemical shift...
-
The 1H NMR spectrum of compound A (C8H8O) consists of two singlets of equal area at 5.1 (sharp) and 7.2 ppm (broad). On treatment with excess hydrogen bromide, compound A is converted to a single...
-
A 100-m-long conductor of uniform cross section has a voltage drop of 4 V between its ends. If the density of the current flowing through it is 1.4 x 106 (A/m2), identify the material of the...
-
1. As a strategic leader, what error might Mary Briggs be making? 2. To what extent should a school superintendent be spending more time on strategy than operational problems? 3. What advice might...
-
A triangular fin of stainless steel (18% Cr, 8% Ni) is attached to a plane wall maintained at 460C. The fin thickness is 6.4 mm, and the length is 2.5 cm. The environment is at 93C, and the...
-
Why would anyone design a tuned absorber for a machine?
-
The town of Newport obtained a corporate MasterCard that was given to the town clerk for purchasing fuel for the town hall. The town clerk used the card for personal restaurant, hotel, and gift shop...
-
Sweet Catering completed the following selected transactions during May 2016: May 1: Prepaid rent for three months, $1,200 May 5: Received and paid electricity bill, $130 May 9: Received cash for...
-
Tourist gaze and choice of destination What comes to mind when a person thinks of a holiday destination? Through time, holiday destinations become famous for a specific holiday experience. Choice of...
-
How would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers? (a) C and 2 (b) 2CH2CH2CO2H CH3CH(CO2H)2 and (c) CH3CH2CH2C02H and H-CH2CH2CH and -2 (d)...
-
Propose a structure for a compound C4H7N that has the following IR and 1H NMR spectra: 100 80 - 60 40 - 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) TMS O ppm 10 9. 8. 3. Chemical...
-
Define overhead costs. How are they allocated?
-
Note that the following guidance is the minimum level of response required. Analyse: when a question asks you to analyse something, you should do so in detail, and identify important points and key...
-
Using the state pattern, you have to implement a robot that can; walk, walk slowly, walk fast, and cook based on an action provided by the user. In your design take the following into consideration:...
-
Can companies reconcile corporate impacts on people, planet and prosperity with satisfying their fundamental mission to achieve profit and shareholder returns? 2. SDGs are becoming increasingly...
-
Marriot Hotels and Resorts have decided to upgrade their entire Hotel Management Network and Application Infrastructure across 5 cities in Ontario. As part of this entire RFP procurement process, the...
-
Universal Camera, a camera manufacturer, sold 50 single lens reflex cameras to Lens Master, a retail store that sells cameras. The purchase price was $200 per camera. The written contract between...
-
When using the "longest life" planning horizon, what issue(s) might you have to consider for alternatives whose cash flow profiles are shorter than the "longest life'? a. Determination of salvage...
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
Platinum crystallizes with the face-centered cubic unit cell. The radius of a platinum atom is 139 pm. Calculate the edge length of the unit cell and the density of platinum in g/cm 3 .
-
For which of the following ions does the formal charge give a fairly accurate picture of where the charge really is? Explain in each case. (a) NH4 (b) H3O: (C) NH2 (d) CH3
-
Is there an unbranched alkane containing 23 hydrogen atoms? If so, give its structural formula; if not, explain why not.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
Higgins Company began operations last year. You are a member of the management team investigating expansion ideas that will require borrowing funds from banks. On January 1 , the start of the current...
-
On April 1, 2021, Windsor, Inc. purchased $615,000 of 6% bonds for $639,225 plus accrued interest as an available-for-sale security. Interest is paid on July 1 and January 1 and the bonds mature on...
-
Use the graphical method for linear programming to find the optimal solution for the following problem. Minimize C=3x+15y subject to 2x + 4y 12 5x + 2y 10 and x 0, y 0.

Study smarter with the SolutionInn App


