How would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers?
Question:
How would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers?
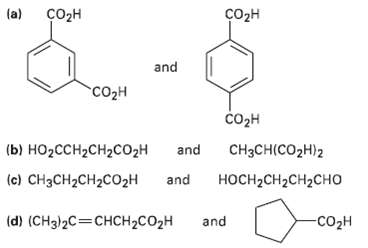
Transcribed Image Text:
(a) созн Cоон and со2н соон (b) НО2СCH2CH2CO2H CH3CH(CO2H)2 and (c) CH3CH2CH2C02H and носH-CH2CH2CHО and -со2н (d) (CH3)2C=CHCH2CO2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
In all of these pairs different numbers of peaks occur in the spectra of each iso...View the full answer

Answered By

Anoop V
I have five years of experience in teaching and I have National Eligibility in teaching (UGC-NET) .
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you distinguish between the following pairs of isomers by 1H NMR spectroscopy? a. CH3CCl3 and CH2ClCHCl2 b. CH3CH2CH2OH and (CH3)2CHOH c d. 0 C-OCH and H C OCH2CH CH2-CH=O and C CH
-
How would you use 1 H NMR spectroscopy to distinguish between the following compounds? (a) (b) (c) (d) (e) (f) . . . CI
-
Distinguish between the following pairs of terms: a. Debt securities classified as held to maturity versus available for sale b. Equity securities classified as trading securities versus available...
-
Write a critical review paper on the topic of financial management in the broad sense.
-
A wheelchair user is conducting a job search to land a middle-management position. Given that so many companies are attempting to build a more culturally diverse group of managers, should this job...
-
Refer to the VAR regression results given in the following table. From the various F tests reported in the three regressions given there, what can you say about the nature of causality in the three...
-
State the form of the Laplace equation in axisymmetric spherical coordinates. Verify that the following functions satisfy this equation: \[r \cos \theta ; \quad \cos \theta / r^{2}\] A linear...
-
Mr. Wellington has prepared the following list of statements about service companies and merchandisers. 1. Measuring net income for a merchandiser is conceptually the same as for a service company....
-
It is May 2020. Marge, age 80, was recently widowed and is of sound mind. She was quite shocked by the sudden death of her husband, Gus, due to Covid-19 virus and realizes that life is precious....
-
1. Search Facebook for Sedona, Arizona. Examine a variety of Sedona-area pages that you find. Using the knowledge of this chapter and your personal social media experience, evaluate these pages and...
-
What spectroscopic method could you use to distinguish among the following three isomeric acids? Tell what characteristic features you would expect for eachacid. CH3(CH213CO2H (CH/2CHCH-cO2H...
-
Compound A, C 4 H 8 O 3 , has infrared absorptions at 1710 and 2500 to 3100 cm ?1 and has the 1 H NMR spectrum shown. Propose a structure for A. 11.18 TMS 10 O ppm 8. 6. 3. Chemical shift (6)...
-
Consider the two nonpolar hydrocarbon molecules, both of formula C 5 H 12 . Having exactly the same formula, they have exactly the same number of electrons, yet pentane has a higher boiling point...
-
Describe the materials in the environment, Real Objects versus Open-Ended Materials
-
Kevin, CPA is performing on audit on RST Company. Kevin decides to sample the additions to the equipment account. Kevin determines that the population size is 100 transactions and 25 of these...
-
What will help managers further distinguish product costs? Explain.
-
1. What is a "Bull Market?" 2. What got so many people interested in buying and selling stocks? 3. Why did the stock market rise so rapidly between March 1928 and September 1929? 4. How could average...
-
what literary methods were used within the oracle. What is the main message of the oracle?
-
Write a program in \(\mathrm{R}\) that simulates 1000 samples of size \(n\) from a Pois\(\operatorname{SON}(\theta)\) distribution, where \(n\) and \(\theta\) are specified below. For each sample...
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
Molybdenum crystallizes with the body-centered unit cell. The radius of a molybdenum atom is 136 pm. Calculate the edge length of the unit cell and the density of molybdenum.
-
Draw a Lewis structure for acetonitrile, C2H3N, assuming that all bonding obeys the octet rule, and that no atom bears a formal charge. Acetonitrile contains a carbon-nitrogen triple bond.
-
Compute the formal charges on atom of the following structure. what is the charge on the entire structure? (a) :0: :0 P- :O:
-
Compute the formal charges on atom of the following structure. what is the charge on the entire structure? (a) :0: :0 P- :O:
-
How do you prioritize features and attributes when designing a new product or service? What steps should be taken to ensure that the design process aligns with the organization's overall strategy and...
-
Information that does not have an adjustment reason code to route payments into the correct bank accounts to relay about patient benefit coverage to indicate the amount being paid and the date of...
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...

Study smarter with the SolutionInn App


