Computation of the Prandtl numbers for gases at low density. (a) By using the Eucken formula and
Question:
Computation of the Prandtl numbers for gases at low density.
(a) By using the Eucken formula and experimental heat capacity data, estimate the Prandtl number at 1atm and 300K for each of the gases listed in the table.
(b) For the same gases, compute the Prandtl number directly by substituting the following values of the physical properties into the defining formula Pr = Cpµ/k, and compare the values with the results obtained in (a). All properties are given at low pressure and 300K.
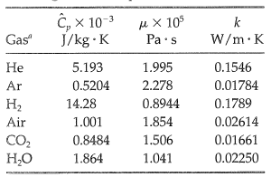
Transcribed Image Text:
t, x 10 μχ 10 J/kg · K Gas Pa·s W/m·K Не 5.193 1.995 0.1546 0.5204 2.278 0.01784 Ar H2 14.28 0.8944 0.1789 1.854 0.02614 Air 1.001 CO, но 1.506 0.01661 0.8484 1.864 1.041 0.02250
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (18 reviews)
Computation of the Prandtl number for gases at low density Us...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Substituting appropriate values for x and y into the formula of Theorem 1.9, show that (a) (b) (c) (-10
-
For liquid metals with Prandtl numbers much less the unity, the hydrodynamic boundary layer is much thinner than the thermal boundary layer. As a result, one may assume that the velocity in the...
-
The density values in Table 12.1 are listed in increasing order. A chemistry student notices that the first four chemical elements that are included are also listed in order of increasing atomic...
-
Data Corporation has four employees and provides group term life insurance coverage for all four employees. Coverage is nondiscriminatory and is as follows: a. How much may Data Corporation deduct...
-
Wegesin and Stem (2004) found greater consistency (less variability) in the memory performance scores for younger women than for older women. The following data represent memory scores obtained for...
-
Sketch the graph of y = y = x1/3. Obviously, its only x-intercept is zero. Convince yourself that Newton's Method fails to converge. Explain this failure.
-
Consider the plot of power as a function of effect size for the two-sample t-test shown in Figure 4.3. a. Create a plot to show power as a function of both effect size and sample size while keeping...
-
Warner Motors's stock is trading at $20 a share. Call options that expire in three months with a strike price of $20 sell for $1.50. Which of the following will occur if the stock price increases...
-
Helix Corporation uses the weighted - average method in its process costing system. It produces prefabricated flooring in a series of steps carried out in production departments. All of the material...
-
The position of a particle as a function of time is given by r(vector) = (5.0i + 4.0j)t 2 m, where t is in seconds. a. What is the particles distance from the origin at t = 0, 2, and 5 s? b. Find an...
-
Prediction of thermal conductivities of gases at tow density, (a) Compute the thermal conductivity of argon at 100C and atmospheric pressure, using the Chapman-Enskog theory and the Lennard-Jones...
-
Estimation of the thermal conductivity of a dense gas, predict the thermal conductivity of methane at 110.4atm and 127F by the following methods: (a) Use Fig. 9.2-1. Obtain the necessary critical...
-
Why might a big company like Lenovo want to develop strategic partnerships with locally owned computer stores? Describe what Lenovo would have to do to maintain such relationships.
-
The local dry cleaner is concerned about the risks from its use of chemicals, which evaporate into the air and occasionally are spilled onto the ground. The cleaner is concerned about liability for...
-
What continuous uniform series of cash flows over 7 years can be paid out of a fund having $20,000 at the present (or time 0) if interest is 10 percent compounded continuously?
-
If a potential victim can take precautions to avoid an accident (such as looking both ways before crossing a street), which liability system yields more efficient levels of precaution, strict...
-
Describe how a tax scheme could be used to regulate automobile pollution in an urban area.
-
In the early 1990 s, the chief economist of the World Bank came under a great deal of criticism for suggesting that developing countries take advantage of opportunities to import hazardous and...
-
Monty (Idris Elba), a hardworking automobile mechanic, loses custody of his three daughters to his evil ex-wife, Jennifer (Tasha Smith). He begins working as a chauffeur for Julia (Gabriele Union),...
-
Should we separate the debt and equity features of convertible debt? Team 1: Pro separation: Present arguments in favor of separating the debt and equity features of convertible debt. Team 2: Against...
-
What reagent solution might you use to separate the cations in each of the following mixtures? (a) PbSO 4 (s) and Cu(NO 3 ) 2 (s) (b) Mg(OH) 2 (s) and BaSO 4 (s) (c) PbCO 3 (s) and CaCO 3 (s)
-
The chapter opener suggests that deficits are generally a problem for countries. Based on the models presented in Section 13.3, are there circumstances in which it might be possible that deficits...
-
Use Equation (13.4), repeated below, to demonstrate how an increase in the budget deficit must increase the trade deficit if neither consumption nor investment changes. What must happen if there is...
-
Explain the reasons for the projected increase in the U.S. budget deficit in coming years.
-
Consider the vectors u=2ij2k and v=2i+2j+4k. Find the vector components of u that are parallel and perpendicular to v.
-
1. Determine whether the following sets form subspaces and justify your answers. (a) {(1, 2): 122 = 0} in R (b) {(1, 2) |1|-|x2|} in R2 (c) {(x1, x2, x3): x3 = 21 or x3 = 12} in R3 (d) {(x1, x2, x3):...
-
Solve the system of two equations for y two equations are: A1=ax+by A2=cx+dy

Study smarter with the SolutionInn App


